Gnaiger 2014 MitoPathways
|
Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. Oroboros MiPNet Publications, Innsbruck:80 pp. — see 5th edition: Gnaiger 2020 BEC MitoPathways. |
Gnaiger Erich (2014) MiPNet
Abstract:
»ISBN 978-3-9502399-8-0 »Open Access«
• Keywords: Q-junction, Respiratory states, Flux control ratios
• O2k-Network Lab: AT Innsbruck Gnaiger E, AT Innsbruck Oroboros
Bioblast online references and notes
- Preface - the 2014 edition of the 'Blue Book' was released at MiP2014 and IOC95.
- References Preface
- Gnaiger E ed (2007) Mitochondrial pathways and respiratory control. 1st ed. Oroboros MiPNet Publications, Innsbruck:96 pp. »Bioblast link«
Chapter 1. Real-time OXPHOS analysis
References OXPHOS analysis
- Altmann R (1894) Die Elementarorganismen und ihre Beziehungen zu den Zellen. Zweite vermehrte Auflage. Verlag Von Veit & Comp, Leipzig:160 pp, 34 Tafeln. - »Bioblast link«
- Dawson KD, Baker DJ, Greenhaff PL, Gibala MJ (2005) An accute decrease in TCA cycle intermediates does not affect aerobic energy delivery in contracting rat skeletal muscle. J Physiol 565:637-43. - »Bioblast link«
- Garlid KD, Semrad C, Zinchenko V (1993) Does redox slip contribute significantly to mitochondrial respiration? In: Schuster S, Rigoulet M, Ouhabi R, Mazat J-P (eds) Modern Trends in Biothermokinetics. Plenum Press, New York, London:287-93. - »Bioblast link«
- Gibala 1998 Am J Physiol Endocrinol Metab|Gibala MJ, MacLean DA, Graham TE, Saltin B (1998) Tricarboxylic acid cycle intermediate pool size and estimated cycle flux in human muscle during exercise. Am J Physiol Endocrinol Metab 275:E235-42. - »Bioblast link« - Concentrations of TCA cycle intermediates.
- Gnaiger E (1983) Heat dissipation and energetic efficiency in animal anoxibiosis. Economy contra power. J Exp Zool 228:471-90. - »Bioblast link«
- Gnaiger E (1993) Efficiency and power strategies under hypoxia. Is low efficiency at high glycolytic ATP production a paradox? In: Surviving Hypoxia: Mechanisms of Control and Adaptation. Hochachka PW, Lutz PL, Sick T, Rosenthal M, Van den Thillart G (eds) CRC Press, Boca Raton, Ann Arbor, London, Tokyo:77-109. - »Bioblast link« - The Gibbs force of phorphorylation of ADP to ATP is FATP = 52 to 66 kJ/mol ATP under intracellular conditions.
- Gnaiger E (1993) Nonequilibrium thermodynamics of energy transformations. Pure Appl Chem 65:1983-2002. - »Bioblast link«
- Gnaiger E (2003) Oxygen conformance of cellular respiration. A perspective of mitochondrial physiology. Adv Exp Med Biol 543:39-55. - »Bioblast link«
- Gnaiger E, Kuznetsov AV (2002) Mitochondrial respiration at low levels of oxygen and cytochrome c. Biochem Soc Trans 30:252-8. - »Bioblast link«
- Gnaiger E, Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Steurer W, Margreiter R (2000b) Mitochondria in the cold. In: Life in the cold. (Heldmaier G, Klingenspor M, eds) Springer, Heidelberg, Berlin, New York:431-42. - »Bioblast link« – MiR05 as the basis of MiR06.
- Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. Biochim Biophys Acta 1365:249-54. - »Bioblast link«
- Gnaiger E, Lassnig B, Kuznetsov AV, Rieger G, Margreiter R (1998) Mitochondrial oxygen affinity, respiratory flux control, and excess capacity of cytochrome c oxidase. J Exp Biol 201:1129-39. - »Bioblast link«
- Gueguen N, Lefaucheur L, Ecolan P, Fillaut M, Herpin P (2005) Ca2+-activated myosin-ATPases, creatine and adenylate kinases regulate mitochondrial function according to myofibre type in rabbit. J Physiol 564:723-35. - »Bioblast link«
- Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer-pathway. XLII. Reconstitution of the electron transfer-pathway. J Biol Chem 237:2661-9. - »Bioblast link«
- Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Mark W, Steurer W, Saks V, Usson Y, Margreiter R, Gnaiger E (2004) Mitochondrial defects and heterogeneous cytochrome c release after cardiac cold ischemia and reperfusion. Am J Physiol Heart Circ Physiol 286:H1633–41. - »Bioblast link« – Cytochrome ‘’c’’ test.
- Lemieux H, Garedew A, Blier PU, Tardif J-C, Gnaiger E (2006) Temperature effects on the control and capacity of mitochondrial respiration in permeabilized fibers of the mouse heart. Biochim Biophys Acta, EBEC Short Reports Suppl 14:201-2. - »Bioblast link«
- Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191:144-8. - »Bioblast link«
- Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Glynn Research Ltd, Bodmin:192 pp. - The Grey Book 1.
- Mitchell P (1968) Chemiosmotic coupling and energy transduction. Glynn Research Ltd, Bodmin:111 pp. - The Grey Book 2.
- Mitchell P, Moyle J (1967) Respiration-driven proton translocation in rat liver mitochondria. Biochem J 105:1147-62. - »Bioblast link«
- Mootha VK, Arai AE, Balaban RS (1997) Maximum oxidative phosphorylation capacity of the mammalian heart. Am J Physiol 272:H769-75. - »Bioblast link« – [Pi] <10 mM and [ADP] <0.4 mM limit OXPHOS in isolated heart mitochondria.
- Nicholson JK, Holmes E, Kinross JM, Darzi AW, Takats Z, Lindon JC (2012) Metabolic phenotyping in clinical and surgical environments. Nature 491:384-92. - »Bioblast link«
- Owen OE, Kalhan SC, Hanson RW (2002) The key role of anaplerosis and cataplerosis for citric acid cycle function. J Biol Chem 277:30409-12. - »Bioblast link«
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopsies of human muscle. Methods Mol Biol 810:25-58. - »Bioblast link« - >90% saturation is reached only >5 mM ADP, yet few studies use such high [ADP] in permeabilized tissues and cells. - Oxygen limitation of respiration below air saturation.
- Puchowicz MA, Varnes ME, Cohen BH, Friedman NR, Kerr DS, Hoppel CL (2004) Oxidative phosphorylation analysis: assessing the integrated functional activity of human skeletal muscle mitochondria – case studies. Mitochondrion 4:377-85. - »Bioblast link« - Cytochrome c test.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. Mol Cell Biochem 208:37-44. - »Bioblast link« - Cytochrome c test.
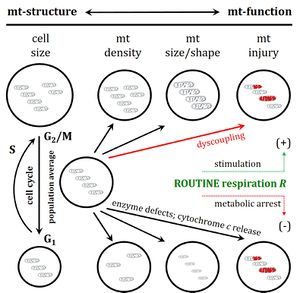
- Renner K , Amberger A, Konwalinka G, Gnaiger E (2003) Changes of mitochondrial respiration, mitochondrial content and cell size after induction of apoptosis in leukemia cells. Biochim Biophys Acta 1642:115-23. - »Bioblast link«
- Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. Biochem J 370:751-62. - »Bioblast link«
- Saks VA, Veksler VI, Kuznetsov AV, Kay L, Sikk P, Tiivel T, Tranqui L, Olivares J, Winkler K, Wiedemann F, Kunz WS (1998) Permeabilised cell and skinned fiber techniques in studies of mitochondrial function in vivo. Mol Cell Biochem 184:81-100. - »Bioblast link« - The apparent Km for ADP increases up to 0.5 mM in some permeabilized muscle fibres.
- Territo PR, Mootha VK, French SA, Balaban RS (2000) Ca2+ activation of heart mitochondrial oxidative phosphorylation: role of the F0/F1-ATPase. Am J Physiol Cell Physiol 278:C423-35. - »Bioblast link«
Notes OXPHOS analysis
Chapter 2. Respiratory states: coupling control
References Respiratory coupling states
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. J Biol Chem 217:383-93. - »Bioblast link«
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. J Biol Chem 217:409-27. - »Bioblast link«
- Chance B, Williams GR (1956) The respiratory chain and oxidative phosphorylation. Adv Enzymol17:65-134. - »Bioblast link«
- Estabrook R (1967) Mitochondrial respiratory control and the polarographic measurement of ADP:O ratios. Meth Enzymol 10:41-7. - »Bioblast link«
- Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. Respir Physiol 128:277-97. - »Bioblast link«
- Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. Biochim Biophys Acta 1365:249-54. - »Bioblast link« - Oxygen kinetics is different in the LEAK state without adenylates (LN) and State 4 (LEAK state with ATP, LN).
- Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. Proc Natl Acad Sci U S A 97:11080-5. - »Bioblast link«
- König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. Biochem J 114:589-96. - 3½ has been suggested to indicate an intermediate mitochondrial energy state somewhere between States 3 and 4. Would, therefore, State 4 be considered as being somewhere between State 3 and 5?
Notes: Coupling states
-
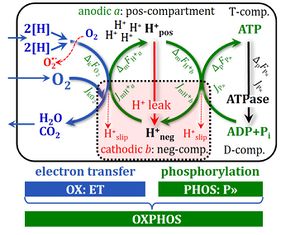
- A colour code is used with red and green in analogy to the states at a traffic light: at red, the motor is running in neutral gear (uncoupled) at minimum turnover without output (producing some heat) just to keep the engine running; at green, the motor is switched into gear and driven in a coupled state with full output. The blue colour is used to indicate a state of maximum input in neutral gear, or pressing fully the accelerator and the clutch simultaneously, which yields maximum turnover without output and produces a maximum of heat. The analogy for coupling in OXPHOS and in cars has its limitations but may help to memories the red/green colour code - you may think of it when your car is in a LEAK at the next red traffic light.
- Proton translocation through pumps is shown by dotted arrows across the inner mt-membrane.
-
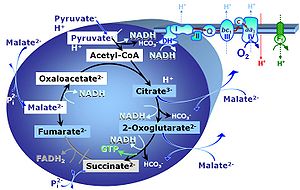
Chapter 3. Mitochondrial pathways to Complex I. Respiratory substrate control with pyruvate, malate and glutamate
References CI
- Brandt U (2006) Energy converting NADH:quinone oxidoreductase (Complex I). Annu Rev Biochem 75:69-92.
- Brewer GJ, Jones TT, Wallimann T, Schlattner U (2004) Higher respiratory rates and improved creatine stimulation in brain mitochondria isolated with antioxidants. Mitochondrion 4:49-57.
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. J Biol Chem 217:409-27. - Substrate depletion in isolated mitochondria is achieved in State 2: ADP is added to induce a transient stimulation of oxygen flux based on oxidation of endogenous substrates.
- Digerness SB, Reddy WJ (1976) The malate-aspartate shuttle in heart mitochondria. J Mol Cell Cardiol. 8:779-85.
- Duchen MR (2004) Roles of mitochondria in health and disease. Diabetes 53, Suppl 1:S96-102. - Mitochondrial glutamate dehydrogenase is particularly active in astrocytes, preventing glutamate induced neurotoxicity.
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837–45. - »Bioblast link«
- Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. Proc Natl Acad Sci U S A 97:11080-5. - »Bioblast link« - Equilibrium ratio of malate to fumarate is 4.1.
- Hildyard JCW, Halestrap AP (2003) Identification of the mitochondrial pyruvate carrier in Saccharomyces cerevidiae. Biochem J 374:607-11.
- Johnson G, Roussel D, Dumas JF, Douay O, Malthiery Y, Simard G, Ritz P (2006) Influence of intensity of food restriction on skeletal muscle mitochondrial energy metabolism in rats. Am J Physiol Endocrinol Metab 291:E460-7. - Uncoupling stimulates coupled OXPHOS respiration, PMP, by 14%.
- Kemp RB, Hoare S, Schmalfeldt M, Bridge CM, Evans PM, Gnaiger E (1994) A thermochemical study of the production of lactate by glutaminolysis and glycolysis in mouse macrophage hybridoma cells. In What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds) Modern Trends in BioThermoKinetics 3, Innsbruck Univ Press:226-31. - Glutamate derived from hydrolyzation of glutamine is a very important aerobic substrate in cultured cells.
- Lemasters JJ (1984) The ATP-to-oxygen stoichiometries of oxidative phosphorylation by rat liver mitochondria. J Biol Chem 259:13123-30. - Malonate added to inhibit the succinate-fumarate reaction exerts only a minor effect on liver mitochondrial respiration.
- Maechler P, Carobbio S, Rubi B (2006) In beta-cells, mitochondria integrate and generate metabolic signals controlling insulin secretion. Int J Biochem Cell Biol 38:696-709.
- Messer JI, Jackman MR, Willis WT (2004) Pyruvate and citric acid cycle carbon requirements in isolated skeletal muscle mitochondria. Am J Physiol Cell Physiol 286:C565-72. - With malate alone and saturating [ADP] isolated rat skeletal muscle mitochondria respire at only 1.3% of OXPHOS capacity with pyruvate+malate. Pyruvate alone yields only 2.1% of OXPHOS capacity (P) with PM.
- Nicholls DG, Ferguson SJ (2002) Bioenergetics 3, Academic Press, London:287 pp. - Carriers.
- Ouhabi R, Boue-Grabot M, Mazat J-P (1994) ATP synthesis in permeabilized cells: Assessment of the ATP/O ratios in situ. In What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds) Modern Trends in BioThermoKinetics 3, Innsbruck Univ Press:141-4. - In fibroblasts, GMP supports a higher respiratory flux than PMP.
- O’Donnell JM, Kudej RK, LaNoue KF, Vatner SF, Lewandowski ED (2004) Limited transfer of cytosolic NADH into mitochondria at high cardiac workload. Am J Physiol Heart Circ Physiol 286:H2237-42.
- Puchowicz MA, Varnes ME, Cohen BH, Friedman NR, Kerr DS, Hoppel CL (2004) Oxidative phosphorylation analysis: assessing the integrated functional activity of human skeletal muscle mitochondria – case studies. Mitochondrion 4:377-85. - OXPHOS with glutamate alone is 50% to 85% of respiration with glutamate&malate. Accumulation of fumarate inhibits succinate dehydrogenase and glutamate dehydrogenase (Caughey et al 1957; Dervartanian, Veeger 1964). - OXPHOS with glutamate&malate is identical or 10% higher than with pyruvate&malate.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. Mol Cell Biochem 208:37-44. - Uncoupling stimulates coupled OXPHOS respiration, PMP, by 15% in human skeletal muscle. OXPHOS with glutamate alone is 50% to 85% of respiration with glutamate&malate. - OXPHOS with glutamate&malate is identical or 10% higher than with pyruvate&malate.
- Thomas et al (2004) - OXPHOS in human skeletal muscle for PMP is 25% higher than for GMP.
- Winkler-Stuck K, Kirches E, Mawrin C, Dietzmann K, Lins H, Wallesch CW, Kunz WS, Wiedemann FR (2005) Re-evaluation of the dysfunction of mitochondrial respiratory chain in skeletal muscle of patients with Parkinson's disease. J Neural Transm 112:499-518. - OXPHOS in human skeletal muscle for PMP is 16% higher than for GMP.
- MitoPedia
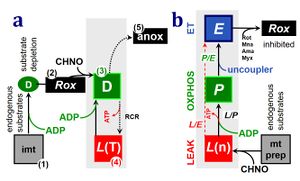
Notes: N-pathway control state - CI-linked
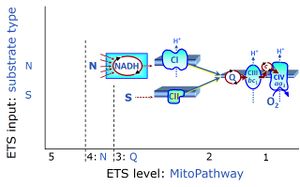
- N-pathway control state
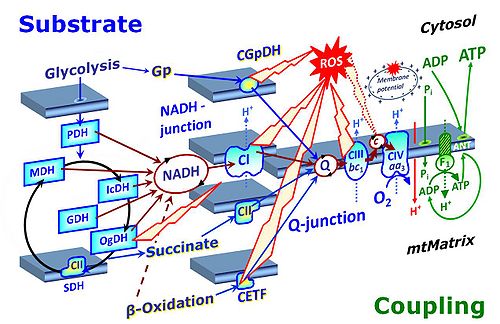
- The metabolic maps in this and the following chapters have been modified and extended in Edition 4 in comparison to previous editions. Added substrates are printed in blue in contrast to intermediates printed in black. CI-linked substrates and intermediates are shown with white background, whereas added succinate and consecutively formed fumarate are distinguished with a yellow background (FADH2 and the corresponding arrows are emphasized by yellow shades). Intermediates with grey background are considered to be present at low concentrations due to metabolite depletion, whereas products with blue background are considered to accumulate in the matrix space or in equilibrium with the large volume of incubation medium or to increase in equilibrium with the supplied substrate.
- Schwerzmann et al (1989) Proc Natl Acad Sci U S A 86:1583-7. - “Of the substrates used here, pyruvate/malate activates the chain at complex I, glutamate/malate and succinate at complexes II and III, ..” - This consideration of glutamate&malate requires correction.
- Ponsot et al (2005) J Cell Physiol 203:479-86. - (a) Respiration (State 3) in permeabilized fibres with malate alone gave 25-50% of the flux with pyruvate+malate. This needs to be discussed in terms of endogenous mitochondrial substrates, which interfere to an unknown degree with the kinetics of respiration after addition of exogenous substrates, or the activity of malic enzyme. (b) Maximal respiration rates in muscle should be evaluated at saturating or high Pi, since at a Pi concentration of 3 mM OXPHOS respiration may be phosphate limited.
- Hulbert et al (2006) J Comp Physiol B 176:93-105. Addition of ‘sparking malate concentrations’. This term can probably be derived from the misconception that tricarboxylic acid cycle intermediates are conserved during respiration of isolated mitochondria. 380 µM malate in conjunction with 2.4 mM pyruvate were used, which makes a comparison difficult between different tissues and different species: the low substrate concentrations may limit PMP flux at various degrees in the different sources of mitochondria, and GMP or PGMP may support higher fluxes than PMP at tissue- and species-specific degrees.
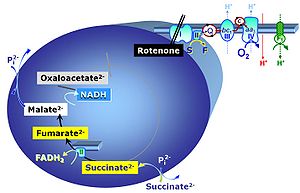
Chapter 4. Mitochondrial pathways to Complex II, glycerophosphate dehydrogenase complex and CETF
References CII
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11. - With succinate alone OXPHOS is 30-40% lower than with succinate+rotenone in human skeletal muscle mitochondria.
- Cecchini G (2003) Function and structure of Complex II of the respiratory chain. Annu Rev Biochem 72:77-109.
- Ernster L, Nordenbrand K (1967) Skeletal muscle mitochondria. In: Estabrook RW, Pullman ME (eds) Meth Enzymol:86-94. – With succinate alone OXPHOS is 30-40% lower than with succinate+rotenone in rat skeletal muscle mitochondria.
- Jackman MR, Willis WT (1996) Characteristics of mitochondria isolated from type I and type IIb skeletal muscle. Am J Physiol Cell Physiol 270:C673-8. - Glycerophosphate oxidation is 10-fold higher in rabbit gracilis mitochondria compared to soleus.
- Lehninger AL (1970) Biochemistry. The molecular basis of cell structure and function Worth:833 pp. - Oxaloacetate is a more potent competitive inhibitor of succinate dehydrogenase than malonate even at small concentration (p 352).
- Muller FL, Liu Y, Abdul-Ghani MA, Lustgarten MS, Bhattacharya A, Jang YC, Van Remmen H (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. Biochem J 409:491–9. - Addition of malate inhibits superoxide production with succinate, probably due to the oxaloacetate inhibition of CII.
- Rauchova H, Drahota Z, Rauch P, Fato R, Lenaz G (2003) Coenzyme Q releases the inhibitory effect of free fatty acids on mitochondrial glycerophosphate dehydrogenase. Acta Biochim Polonica 50:405-13. - Glycerophosphate is an important substrate for respiration in brown adipose tissue mitochondria.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. Mol Cell Biochem 208:37-44. – Glycerophosphate oxidation is relatively slow.
- Sun F, Huo X, Zhai Y, Wang A, Xu J, Su D, Bartlam M, Rao Z (2005) Crystal structure of mitochondrial respiratory membrane protein Complex II. Cell 121:1043–57.
- MitoPedia
Notes CII
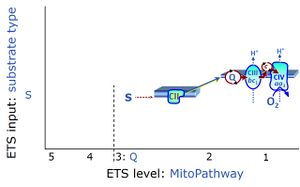
- S-pathway control state
- Ponsot et al (2005) J Cell Physiol 203:479-86. - ‘.. the mitochondrial form of GPDH, which produces FADH2 within the mitochondrial matrix and provides electrons to Compoex II of the phosphorylation chain’. – The mitochondrial glycerophosphate dehydrogenase complex (CGpDH), located on the outer side of the inner mitochondrial membrane, does not provide electrons to CII, but feeds electrons into the Q-cycle entirely independent of CII. FADH2 is not produced within the mitochondrial matrix. Electron transfer takes place from the mitochondrial inner membrane flavoprotein-linked glycerophosphate dehydrogenase complex to CoQ.
Correction
- Page 41 ".. This is similar in mammalian liver, where >10% of malic enzyme is cytosolic. (<10% is erroneously in the present text).
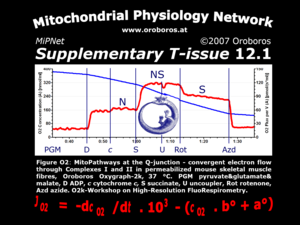
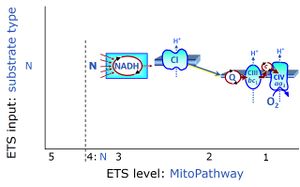
Chapter 5. Mitochondrial pathways to Complexes I&II. Convergent electron transfer at the Q-junction
References CI&II
- Aragonés J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, Dirkx R, Zacchigna S, Lemieux H, Jeoung NH, Lambrechts D, Bishop T, Lafuste P, Diez-Juan A, K Harten S, Van Noten P, De Bock K, Willam C, Tjwa M, Grosfeld A, Navet R, Moons L, Vandendriessche T, Deroose C, Wijeyekoon B, Nuyts J, Jordan B, Silasi-Mansat R, Lupu F, Dewerchin M, Pugh C, Salmon P, Mortelmans L, Gallez B, Gorus F, Buyse J, Sluse F, Harris RA, Gnaiger E, Hespel P, Van Hecke P, Schuit F, Van Veldhoven P, Ratcliffe P, Baes M, Maxwell P, Carmeliet P (2008) Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. Nat Genet 40:170-80. - OXPHOS analysis for phenotyping.
- Bianchi C, Genova ML, Parenti Castelli G, Lenaz G (2004) The mitochondrial respiratory chain is partially organized in a supercomplex assembly: kinetic evidence using flux control analysis. J Biol Chem 279:36562-9.
- Boushel R, Gnaiger E, Schjerling P, Skovbro M, Kraunsoe R, Flemming D (2007) Patients with Type 2 Diabetes have normal mitochondrial function in skeletal muscle. Diabetologia 50:790-6.
- Boushel R, Gnaiger E, Calbet JA, Gonzalez-Alonso J, Wright-Paradis C, Sondergaard H, Ara I, Helge JW, Saltin B (2011) Muscle mitochondrial capacity exceeds maximal oxygen delivery in humans. Mitochondrion 11:303-7.
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11. - CI&II substrate combination.
- Chance B (1965) Reaction of oxygen with the respiratory chain in cells and tissues. J Gen Physiol 49:163-88. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Costa LE, Boveris A, Koch OR, Taquini AC (1988) Liver and heart mitochondria in rats submitted to chronic hypobaric hypoxia. Am J Physiol Cell Physiol 255:C123-C9.
- Digerness SB, Reddy WJ (1976) The malate-aspartate shuttle in heart mitochondria. J Mol Cell Cardiol. 8:779-85.
- Eberhart K, Rainer J, Bindreither D, Ritter I, Gnaiger E, Kofler R, Oefner PJ, Renner K (2011) Glucocorticoid-induced alterations in mitochondrial membrane properties and respiration in childhood acute lymphoblastic leukemia. Biochim Biophys Acta 1807:719-25.
- Estabrook R (1967) Mitochondrial respiratory control and the polarographic measurement of ADP:O ratios. Meth Enzymol 10:41-7.
- Garait B, Couturier K, Servais S, Letexier D, Perrin D, Batandier C, Rouanet J-L, Sibille B, Rey B, Leverve X, Favier R (2005) Fat intake reverses the beneficial effects of low caloric intake on skeletal muscle mitochondrial H2O2 production. Free Radic Biol Med 39:1249–61. - GMP/GMSP substrate control ratio in skeletal muscle of rats fed on various diets ranges from 0.7 to 0.8.
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837–45. - »Bioblast link«
- Gnaiger E, Wright-Paradis C, Sondergaard H, Lundby C, Calbet JA, Saltin B, Helge J, Boushel R (2005) High-resolution respirometry in small biopsies of human muscle: correlations with body mass index and age. Mitochondr Physiol Network 10.9:14-5. MiP2005.
- González-Flecha B, Cutrin JC, Boveris A (1993) Time course and mechanism of oxidative stress and tissue damage in rat liver subjected to in vivo ischemia-reperfusion. J Clin Invest 91:456-64. - Respiration was measured in states GSP and GMP.
- Gutman M, Coles CJ, Singer TP, Casida JE (1971) On the functional organization of the respiratory chain at the dehydrogenase-coenzyme Q junction. Biochemistry 10:2036-43.
- Hansford RG, Hogue BA, Mildaziene V (1997) Dependence of H2O2 formation by rat heart mitochondria on substrate availability and donor age. J Bioenerg Biomembr 29:89–95.
- Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer-pathway. XLII. Reconstitution of the electron transfer-pathway. J Biol Chem 237:2661-9.
- König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. Biochem J 114:589-96.
- Krebs HA (1935) CXCVII. Metabolism of amino-acids. III. Deamination of amino-acids. Biochem J 29:1620-44.
- Kunz WS, Kudin A, Vielhaber S, Elger CE, Attardi G, Villani G (2000) Flux control of cytochrome c oxidase in human skeletal muscle. J Biol Chem 275:27741-5.
- Kuznetsov AV, Clark JF, Winkler K, Kunz WS (1996) Increase of flux control of cytochrome c oxidase in copper-deficient mottled brindled mice. J Biol Chem 271:283-8.
- Kuznetsov AV, Strobl D, Ruttmann E, Königsrainer A, Margreiter R, Gnaiger E (2002) Evaluation of mitochondrial respiratory function in small biopsies of liver. Analyt Biochem 305:186-94. - S(Rot) alone supports a higher flux than GM in liver mitocondria.
- Kuznetsov AV, Winkler K, Kirches E, Lins H, Feistner H, Kunz WS (1997) Application of inhibitor titrations for the detection of oxidative phosphorylation defects in saponin-skinned muscle fibers of patients with mitochondrial diseases. Biochim Biophys Acta 1360:142-50. - OXPHOS with glutamate&malate is identical or 10% higher than with pyruvate&malate.
- LaNoue KF, Bryla J, Williamson JR (1972) Feedback interactions in the control of citric acid cycle activity in rat heart mitochondria. J Biol Chem 247:667-79.
- LaNoue KF, Schoolwerth AC (1979) Metabolite transport in mitochondria. Annu Rev Biochem 48:871-922.
- Lehninger AL (1970) Biochemistry. The molecular basis of cell structure and function Worth:833 pp. - Electron transport chain.
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - »Bioblast link«
- Lemieux H, Semsroth S, Antretter H, Hoefer D, Gnaiger E (2011) Mitochondrial respiratory control and early defects of oxidative phosphorylation in the failing human heart. Int J Biochem Cell Biol 43:1729–38. - »Bioblast link« - SUIT protocols.
- Lenaz G, Genova ML (2009) Structural and functional organization of the mitochondrial respiratory chain: A dynamic super-assembly. Int J Biochem Cell Biol 41:1750-72.
- Llesuy S, Evelson P, González-Flecha B, Peralta J, Carreras MC, Poderoso JJ, Boveris A (1994) Oxidative stress in muscle and liver of rats with septic syndrome. Free Radic Biol Med 16:445-51. - GMP/GSP substrate control ratios are 0.8 and 0.7 in liver mitochondria (male Wistar and female Sprague-Dawley, respectively).
- Mogensen M, Sahlin K (2005) Mitochondrial efficiency in rat skeletal muscle: influence of respiration rate, substrate and muscle type. Acta Physiol Scand 185:229-36. - The flux control ratio of palmitoylcarnitine&malate/PM (OXPHOS) is 0.6 for mitochondria isolated from rat extensor digitorum longus muscle (mainly type II fibre type), but is 0.95 in rat soleus muscle (type I fibre type).
- Muller FL, Liu Y, Abdul-Ghani MA, Lustgarten MS, Bhattacharya A, Jang YC, Van Remmen H (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. Biochem J 409:491–9.
- Nicholls DG, Ferguson SJ (2002) Bioenergetics 3, Academic Press, London:287 pp. - Electron transport chain.
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopisies of human muscle. Methods Mol Biol 810:25-58. - »Bioblast link« - SUIT protocols.
- Pesta D, Hoppel F, Macek C, Messner H, Faulhaber M, Kobel C, Parson W, Burtscher M, Schocke M, Gnaiger E (2011) Similar qualitative and quantitative changes of mitochondrial respiration following strength and endurance training in normoxia and hypoxia in sedentary humans. Am J Physiol Regul Integr Comp Physiol 301:R1078–87. - »Bioblast link« - SUIT protocols.
- Rasmussen HN, Rasmussen UF (1997) Small scale preparation of skeletal muscle mitochondria, criteria for integrity, and assays with reference to tissue function. Mol Cell Biochem 174:55-60.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. Mol Cell Biochem 208:37-44. - OXPHOS with palmitoylcarnitine&malate is 0.60 of OXPHOS with GM.
- Rasmussen UF, Rasmussen HN, Krustrup P, Quistorff B, Saltin B, Bangsbo J (2001) Aerobic metabolism of human quadriceps muscle: in vivo data parallel measurements on isolated mitochondria. Am J Physiol Endocrinol Metab 280:E301–7.
- Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. Biochem J 370:751-62.
- Rumsey WL, Schlosser C, Nuutinen EM, Robiolio M, Wilson DF (1990) Cellular energetics and the oxygen dependence of respiration in cardiac myocytes isolated from adult rat. J Biol Chem 265:15392-9. - 2.5-fold increase of respiration in the LEAK state with GMS compared to GM.
- Schägger H, Pfeiffer K (2001) The ratio of oxidative phosphorylation complexes I-V in bovine heart mitochondria and the composition of respiratory chain supercomplexes. J Biol Chem 276:37861-7.
- Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS (2005) Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A 102:5618-23.
- Simon J, van Spanning RJM, Richardson DJ (2008) The organisation of proton motive and non-proton motive redox loops in prokaryotic respiratory systems. Biochim Biophys Acta 1777:1480–90.
- Steinlechner-Maran R, Eberl T, Kunc M, Margreiter R, Gnaiger E (1996) Oxygen dependence of respiration in coupled and uncoupled endothelial cells. Am J Physiol Cell Physiol 271:C2053-61.
- Steinlechner-Maran R, Eberl T, Kunc M, Schröcksnadel H, Margreiter R, Gnaiger E (1997) Respiratory defect as an early event in preservation/reoxygenation injury in endothelial cells. Transplantation 63:136-42.
- Stadlmann S, Rieger G, Amberger A, Kuznetsov AV, Margreiter R, Gnaiger E (2002) H2O2-mediated oxidative stress versus cold ischemia-reperfusion: mitochondrial respiratory defects in cultured human endothelial cells. Transplantation 74:1800-3.
- Sugano T, Oshino N, Chance B (1974). Mitochondrial functions under hypoxic conditions. The steady states of cytochrome c reduction and energy metabolism. Biochim Biophys Acta 347:340-58. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Tonkonogi M, Walsh B, Tiivel T, Saks V, Sahlin K (1999) Mitochondrial funciton in human skeletal muscle is not impaired by high intensity exercise. Eur J Physiol 437:562-8.
- Torres NV, Mateo F, Sicilia J, Meléndez-Hevia E. (1988) Distribution of the flux control in convergent metabolic pathways: theory and application to experimental and simulated systems. Int J Biochem 20:161-5.
- Villani G, Attardi G (1997) In vivo control of respiration by cytochrome c oxidase in wild-type and mitochondrial DNA mutation-carrying human cells. Proc Natl Acad Sci U S A 94:1166-71.
- Villani G, Greco M, Papa S, Attardi G (1998) Low reserve capacity of cytochrome c oxidase capacity in vivo in the respiratory chain of a variety of human cell types. J Biol Chem 273:31829-36.
- Votion DM, Gnaiger E, Lemieux H, Mouithys-Mickalad A, Serteyn D (2012) Physical fitness and mitochondrial respiratory capacity in horse skeletal muscle. PLoS One 7:e34890. - »Bioblast link« - SUIT protocols.
- Wiedemann FR, Winkler K, Kuznetsov AV, Bartels C, Vielhaber S, Feistner H, Kunz WS (1998) Impairment of mitochondrial function in skeletal muscle of patients with amyotrophic lateral sclerosis. J Neurol Sci 156:65-72. - OXPHOS with glutamate&malate is identical or 10% higher than with pyruvate&malate.
- Wilson DF, Rumsey WL, Green TJ, Vanderkooi J (1988). The oxygen dependence of mitochondrial oxidative phosphorylation measured by a new optical method for measuring oxygen concentration. J Biol Chem 263:2712-8. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Zoccarato F, Cavallini L, Bortolami S, Alexandre A (2007) Succinate modulation of H2O2 release at NADH:ubiquinone oxidoreductase (Complex I) in brain mitochondria. Biochem J 406:125–9. - CI&II substrate combination.
- MitoPedia
Notes: NS-pathway control state - CI&II
- NS-pathway control state
- Identical GMP/GSP or GMP/GMSP ratios of 0.7 are reported for isolated mitochondria (Rasmussen and Rasmussen 2000; Capel et al 2005) and permeabilized fibres (Kunz et al 2000). For a review see Gnaiger (2009).
Chapter 6. Normalization of flux - past the RCR: on efficiency, coupling and substrate control factors
Correction
- Tab. 6.2. ET-capacity of HEK 293 cells is 47 ± 7 (the value of 14 ± 2 given in the table is that of ROUTINE respiration).
References Normalization of flux
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. J Biol Chem 217:383-93.
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. J Biol Chem 217:409-27.
- Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. Respir Physiol 128:277-97. - »Bioblast link«
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopisies of human muscle. Methods Mol Biol 810:25-58. - »Bioblast link« - SUIT protocols.
Chapter 7. Conversions of metabolic fluxes
References Conversions
- Brooks GA, Hittelman KJ, Faulkner JA, Beyer RE (1971) Temperature, skeletal muscle mitochondrial functions, and oxygen debt. Am J Physiol 220:1053-9.
- Gnaiger E (1983) Symbols and units: Toward standardization. In: Polarographic Oxygen Sensors. Aquatic and Physiological Applications. Gnaiger E, Forstner H (eds), Springer, Berlin, Heidelberg, New York:352-8.
- Slater EC, Rosing J, Mol A (1973) The phosphorylation potential generated by respiring mitochondria. Biochim Biophys Acta 292:534-53.
Apendix: Abbreviations
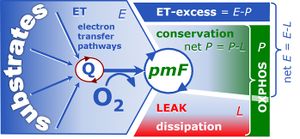
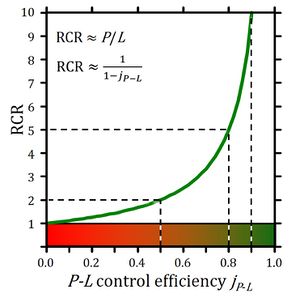
A1. Respiratory coupling states and coupling control ratios
- Correction in Table A1.2. Excess E-R capacity: the symbol should not be ExP but ExR=E-R
A2. Substrates, uncouplers and inhibitors
Follow-up
- The 2007 edition is cited in:
- Muller FL et al (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. Biochem J 409:491–9. - »Bioblast link«
- Gnaiger E (2008) Polarographic oxygen sensors, the oxygraph and high-resolution respirometry to assess mitochondrial function. In: Mitochondrial Dysfunction in Drug-Induced Toxicity (Dykens JA, Will Y, eds) John Wiley:327-52. - »Bioblast link«
- Dröse S, Hanley PJ, Brandt U (2009) Ambivalent effects of diazoxide on mitochondrial ROS production at respiratory chain Complexes I and III. Biochim Biophys Acta 1790:558-65. - »Bioblast link«
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - »Bioblast link«
- Hickey AJ, Chai CC, Choong SY, de Freitas Costa S, Skea GL, Phillips AR, Cooper GJ (2009) Impaired ATP turnover and ADP supply depress cardiac mitochondrial respiration and elevate superoxide in nonfailing spontaneously hypertensive rat hearts. Am J Physiol Cell Physiol 297:C766–74. - »Bioblast link«
- Hoffman DL, Brookes PS (2009) Oxygen sensitivity of mitochondrial reactive oxygen species generation depends on metabolic conditions. J Biol Chem 284:16236-45.
- Sahlin K (2009) Control of lipid oxidation at the mitochondrial level. Appl Physiol Nutr Metab 34:382-8.
- Bazil JN, Buzzard GT, Rundell AE (2010) Modeling mitochondrial bioenergetics with integrated volume dynamics. PLoS Comput Biol. 6:e1000632.
- Renner-Sattler K, Gnaiger E, eds (2010) Mitochondrial physiology. The many functions of the organism in our cells. Mitochondr Physiol Network 15.06:128 pp.
- Scandurra FM, Gnaiger E (2010) Cell respiration under hypoxia: facts and artefacts in mitochondrial oxygen kinetics. Adv Exp Med Biol 662:7-25. - »Bioblast link«
- Sivitz WI, Yorek MA (2010) Mitochondrial dysfunction in diabetes: from molecular mechanisms to functional significance and therapeutic opportunities. Antioxid Redox Signal 12:537-77.
- Sivitz WI (2010) Mitochondrial Dysfunction in Obesity and Diabetes. US Endocrinology 6:20-7.
- Iftikar F, MacDonald JR, Hickey AJ (2010) Thermal limits of portunid crab heart mitochondria: Could more thermo-stable mitochondria advantage invasive species? JEMBE 395:232-9. - »Bioblast link«
- Mittal A, Hickey AJ, Chai CC, Loveday BP, Thompson N, Dare A, Delahunt B, Cooper GJ, Windsor JA, Phillips AR (2011) Early organ-specific mitochondrial dysfunction of jejunum and lung found in rats with experimental acute pancreatitis. HPB (Oxford) 13:332-41. - »Bioblast link«
- Pichaud N, Ballard JW, Tanguay RM, Blier PU (2011) Thermal sensitivity of mitochondrial functions in permeabilized muscle fibers from two populations of Drosophila simulans with divergent mitotypes. Am J Physiol Regul Integr Comp Physiol 301:R48-59. - »Bioblast link«
- Ye F, Lemieux H, Hoppel CL, Hanson RW, Hakimi P, Croniger CM, Puchowicz M, Anderson VE, Fujioka H, Stavnezer E (2011) Peroxisome proliferator-activated receptor γ (PPARγ) mediates a Ski-induced shift from glycolysis to oxidative energy metabolism. J Biol Chem 286:40013-24. - »Bioblast link«
- Cordeiro OD, Silva TS, Alves RN, Costas B, Wulff T, Richard N, de Vareilles M, Conceição LE, Rodrigues PM (2012) Changes in liver proteome expression of Senegalese sole (Solea senegalensis) in response to repeated handling stress. Mar Biotechnol (NY)14:714-29.
- Everett MV, Antal CE, Crawford DL (2012) The effect of short-term hypoxic exposure on metabolic gene expression. J Exp Zool A Ecol Genet Physiol 317:9-23.
- Miquel E, Cassina A, Martinez-Palma L, Bolatto C, Trias E, Gandelman M, Radi R, Barbeito L, Cassina P (2012) Modulation of astrocytic mitochondrial function by dichloroacetate improves survival and motor performance in inherited amyotrophic lateral sclerosis. PLoS One 7:e34776. - »Bioblast link«
- de Paula Martins R, Glaser V, da Luz Scheffer D, de Paula Ferreira PM, Wannmacher CM, Farina M, de Oliveira PA, Prediger RD, Latini A (2013) Platelet oxygen consumption as a peripheral blood marker of brain energetics in a mouse model of severe neurotoxicity. J Bioenerg Biomembr 45:449-57. - »Bioblast link«
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopisies of human muscle. Methods Mol Biol 810:25-58. - »Bioblast link«
- Perry CG, Kane DA, Lanza IR, Neufer PD (2013) Methods for assessing mitochondrial function in diabetes. Diabetes 62:1041-53. - »Bioblast link«
- Chu MJJ, Dare AJ, Bartlett ASJR, Phillips ARJ, Hickey AJ (2013) Effect of hepatic steatosis on bioenergetic function during hepatic ischemia-reperfusion: a aystematic review. The Open Transplantation J 6:10-32.
- Gispert S, Parganlija D, Klinkenberg M, Dröse S, Wittig I, Mittelbronn M, Grzmil P, Koob S, Hamann A, Walter M, Büchel F, Adler T, Hrabé de Angelis M, Busch DH, Zell A, Reichert AS, Brandt U, Osiewacz HD, Jendrach M, Auburger G (2013) Loss of mitochondrial peptidase Clpp leads to infertility, hearing loss plus growth retardation via accumulation of CLPX, mtDNA and inflammatory factors. Hum Mol Genet 22:4871-87. - »Bioblast link«
- Hirzel E, Lindinger PW, Maseneni S, Giese M, Rhein VV, Eckert A, Hoch M, Krähenbühl S, Eberle AN (2013) Differential modulation of ROS signals and other mitochondrial parameters by the antioxidants MitoQ, resveratrol and curcumin in human adipocytes. J Recept Signal Transduct Res 33:304-12. - »Bioblast link«
- Cordero-Reyes AM, Gupte AA, Youker KA, Loebe M, Hsueh WA, Torre-Amione G, Taegtmeyer H, Hamilton DJ (2014) Freshly isolated mitochondria from failing human hearts exhibit preserved respiratory function. J Mol Cell Cardiol doi: 10.1016/j.yjmcc.2013.12.029. - »Bioblast link«
- The 2007 edition is cited in:
- The 2010 Edition is cited in:
- Chung et al (2011) J Innov Opt Health Sci 4:361–72.
- Darrah-Wiedemeier KL (2015) The physiological effects of the seed treatment Stamina on maize seedling length, ADP:O ratios, and respiration rates. Graduate Theses and Dissertations Paper 14673:55 pp. - »Bioblast link«
- Granados-Castro LF, Rodríguez-Rangel DS, Montaño M, Ramos C, Pedraza-Chaverri J (2013) Wood smoke exposure induces a decrease in respiration parameters and in the activity of respiratory complexes I and IV in lung mitochondria from guinea pigs. Environ Toxicol. doi: 10.1002/tox.21922.
- Maldonado F, Packard TT, Gómez M (2012) Understanding tetrazolium reduction and the importance of substrates in measuring respiratory electron transport activity. J Exp Marine Biol Ecol 434–435:110–18.
- Widmann M (2014) Effekte von Noradrenalin und LPS auf die mitochondriale Atmung von Alveolarmakrophagen. Thesis Universität Ulm, Medizinische Fakultät. 63 pp.
- The 2010 Edition is cited in:
- The 2012 edition is cited in:
- Castelein N, Muschol M, Dhondt I, Cai H, De Vos WH, Dencher NA, Braeckman BP (2014) Mitochondrial efficiency is increased in axenically cultured Caenorhabditis elegans. Exp Gerontol. - »Bioblast link«
- Gnaiger E, Meissner B, Laner V, eds (2012) Bioblast 2012: Mitochondrial Competence. Mitochondr Physiol Network 17.12. Oroboros MiPNet Publications, Innsbruck:96 pp. - »Bioblast link«
- Guitart T, Picchioni D, Piñeyro D, Ribas de Pouplana L (2013) Human mitochondrial disease-like symptoms caused by a reduced tRNA aminoacylation activity in flies. Nucleic Acids Res 41:6595-608. - »Bioblast link«
- Karabatsiakis A, Bock C, Salinas-Manrique J, Kolassa S, Calzia E, Dietrich DE, Kolassa I-T (2014) Mitochondrial respiration in peripheral blood mononuclear cells correlates with depressive subsymptoms and severity of major depression. Translational Psychiatry 4:e397. - »Bioblast link«
- Krumschnabel G, Eigentler A, Fasching M, Gnaiger E (2014) Use of safranin for the assessment of mitochondrial membrane potential by high-resolution respirometry and fluorometry. Methods Enzymol 542:163-81. - »Bioblast link«
- Laner V, Bidaurratzaga E, Gnaiger E, eds (2013) Comparative Mitochondrial Physiology. MiP2013. Mitochondr Physiol Network 18.08:96 pp. - »Bioblast link«
- The 2012 edition is cited in:
- The 2014 edition is cited in:
- Burtscher J, Zangrandi L, Schwarzer C, Gnaiger E (2015) Differences in mitochondrial function in homogenated samples from healthy and epileptic specific brain tissues revealed by high-resolution respirometry. Mitochondrion 25:104-12. - »Bioblast link«
- Harrison DK, Fasching M, Fontana-Ayoub M, Gnaiger E (2015) Cytochrome redox states and respiratory control in mouse and beef heart mitochondria at steady-state levels of hypoxia. J Appl Physiol 119:1210-8. - »Bioblast link«
- Laner V, Gnaiger E, eds (2014) Mitochondrial physiology – methods, concepts and biomedical perspectives. MiP2014. Mitochondr Physiol Network 19.13:88 pp. - »Bioblast link«
- Makrecka-Kuka M, Krumschnabel G, Gnaiger E (2015) High-resolution respirometry for simultaneous measurement of oxygen and hydrogen peroxide fluxes in permeabilized cells, tissue homogenate and isolated mitochondria. Biomolecules 5:1319-38. - »Bioblast link«
- Schöpf B, Schäfer G, Weber A, Talasz H, Eder IE, Klocker H, Gnaiger E (2016) Oxidative phosphorylation and mitochondrial function differ between human prostate tissue and cultured cells. FEBS J. - »Bioblast link«
- The 2014 edition is cited in:
Labels: MiParea: Respiration, Instruments;methods, mt-Biogenesis;mt-density, Comparative MiP;environmental MiP, Exercise physiology;nutrition;life style, mt-Medicine, mt-Awareness
Organism: Human, Mouse
Tissue;cell: Heart, Skeletal muscle, Fibroblast
Preparation: Intact cells, Permeabilized cells, Permeabilized tissue, Homogenate, Isolated mitochondria
Regulation: Coupling efficiency;uncoupling, Flux control, mt-Membrane potential, Threshold;excess capacity, Uncoupler Coupling state: LEAK, ROUTINE, OXPHOS, ET Pathway: F, N, S, Gp, CIV, NS, ROX HRR: Oxygraph-2k, O2k-Fluorometer, Theory
MitoPathways