Baglivo 2022 MitoFit-QC
| Baglivo E, Cardoso LHD, Cecatto C, Gnaiger E (2022) Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry. https://doi.org/10.26124/mitofit:2022-0018.v2 — 2022-08-04 published in Bioenerg Commun 2022.8. |
» MitoFit Preprints 2022.18.
Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry ![]()
MitoFit Preprints (2022) MitoFit Prep
Abstract:
- Version 2 (v2) 2022-05-09 doi:10.26124/mitofit:2022-0018.v2
- Version 1 (v1) 2022-05-05 doi:10.26124/mitofit:2022-0018.v1 - »Link to all versions«
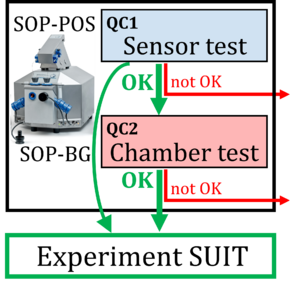
Baglivo 2022 Abstract Bioblast: Evaluation of instrumental reproducibility is a primary component of quality control to quantify the precision and limit of detection of analytical procedures. A pre-analytical instrumental standard operating procedure (SOP) is implemented in high-resolution respirometry consisting of: (1) a daily SOP-POS for air calibration of the polarographic oxygen sensor (POS) in terms of oxygen concentration cO2 [µM]. This is part of the sensor test to evaluate POS performance; (2) a monthly SOP-BG starting with the SOP-POS followed by the chamber test quantifying the instrumental O2 background. The chamber test focuses on the slope dcO2/dt [pmol∙s−1∙mL−1] to determine O2 consumption by the POS and O2 backdiffusion into the chamber as a function of cO2 in the absence of sample. Finally, zero O2 calibration completes the sensor test.
We applied this SOP in a 3-year study using 48 Oroboros O2k chambers. Stability of air and zero O2 calibration signals was monitored throughout intervals of up to 8 months without sensor service. Maximum drift over 1 to 3 days was 0.06 pmol∙s−1∙mL−1, without persistence over time since drift was <0.004 pmol∙s−1∙mL−1 for time intervals of one month, corresponding to a drift per day of 0.2 % of the signal at air saturation. Instrumental O2 background -dcO2/dt was stable within ±1 pmol∙s−1∙mL−1 when measured at monthly intervals. These results confirm the instrumental limit of detection of volume-specific O2 flux at ±1 pmol∙s−1∙mL−1. The instrumental SOP applied in the present study contributes to the generally applicable internal quality control management ensuring the unique reproducibility in high-resolution respirometry.
• O2k-Network Lab: AT Innsbruck Oroboros
ORCID:![]() Baglivo Eleonora
Baglivo Eleonora ![]() Cardoso Luiza HD,
Cardoso Luiza HD, ![]() Cecatto Cristiane,
Cecatto Cristiane, ![]() Gnaiger Erich
Gnaiger Erich
Data availability
- Original files are available Open Access at Zenodo repository: https://doi.org/10.26124/mitofit:2022-0018
References
| Link | Reference | Year | View |
|---|---|---|---|
| Amaral 2021 Nature | Amaral OB, Neves K (2021) Reproducibility: expect less of the scientific paper. Nature 597:329-31. https://doi.org/10.1038/d41586-021-02486-7 | 2021 | PMID: 34526702 Open Access |
| Brookman 2021 Eurachem | Brookman B, Mann I, eds (2021) Eurachem guide: selection, use and interpretation of proficiency testing (PT) schemes (3rd ed). Eurachem Available from www.eurachem.org. | 2021 | Open Access |
| Doerrier 2018 Methods Mol Biol | Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. https://doi.org/10.1007/978-1-4939-7831-1_3 | 2018 | PMID: 29850993 » |
| Gnaiger 2001 Respir Physiol | Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. https://doi.org/10.1016/S0034-5687(01)00307-3 | 2001 | Respir Physiol 128:277-97. PMID: 11718759 |
| Gnaiger 2008 POS | Gnaiger E (2008) Polarographic oxygen sensors, the oxygraph and high-resolution respirometry to assess mitochondrial function. In: Mitochondrial dysfunction in drug-induced toxicity (Dykens JA, Will Y, eds) John Wiley & Sons, Inc, Hoboken, NJ:327-52. | 2008 | |
| Gnaiger 2020 BEC MitoPathways | Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. https://doi.org/10.26124/bec:2020-0002 | 2020 | |
| Gnaiger 2021 MitoFit BCA | Gnaiger E (2021) Bioenergetic cluster analysis – mitochondrial respiratory control in human fibroblasts. MitoFit Preprints 2021.08. https://doi.org/10.26124/mitofit:2021-0008 | 2021 | MitoFit Preprints 2021.08. Bioenergetic cluster analysis – mitochondrial respiratory control in human fibroblasts
|
| Gnaiger 2000 Life in the Cold | Gnaiger E, Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Steurer W, Margreiter R (2000) Mitochondria in the cold. In: Life in the Cold (Heldmaier G, Klingenspor M, eds) Springer, Berlin, Heidelberg:431-42. https://doi.org/10.1007/978-3-662-04162-8_45 | 2000 | |
| Horan 2012 J Gerontol A Biol Sci Med Sci | Horan MP, Pichaud N, Ballard JWO (2012) Review: Quantifying mitochondrial dysfunction in complex diseases of aging. J Gerontol A Biol Sci Med Sci 67:1022-35. https://doi.org/10.1093/gerona/glr263 | 2012 | PMID: 22459622 Open Access |
| Perry 2013 Diabetes | Perry CG, Kane DA, Lanza IR, Neufer PD (2013) Methods for assessing mitochondrial function in diabetes. Diabetes 62:1041-53. https://doi.org/10.2337/db12-1219 | 2013 | PMID: 23520284 Open Access |
| Pesta 2012 Methods Mol Biol | Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods Mol Biol 810:25-58. https://doi.org/10.1007/978-1-61779-382-0_3 | 2012 | PMID: 22057559 |
| Schmidt 2021 J Biol Chem | Schmidt CA, Fisher-Wellman KH, Neufer PD (2021) From OCR and ECAR to energy: perspectives on the design and interpretation of bioenergetics studies. J Biol Chem 207:101140. https://doi.org/10.1016/j.jbc.2021.101140 | 2021 | PMID: 34461088 Open Access |
| Yepez 2018 PLOS One | Yépez VA, Kremer LS, Iuso A, Gusic M, Kopajtich R, Koňaříková E, Nadel A, Wachutka L, Prokisch H, Gagneur J (2018) OCR-Stats: Robust estimation and statistical testing of mitochondrial respiration activities using Seahorse XF Analyzer. https://doi.org/10.1371/journal.pone.0199938 | 2018 | PLOS ONE 13:e0199938. PMID: 29995917 Open Access |
| Zdrazilova 2022 PLOS ONE | Zdrazilova L, Hansikova H, Gnaiger E (2022) Comparable respiratory activity in attached and suspended human fibroblasts. PLoS ONE 17:e0264496. https://doi.org/10.1371/journal.pone.0264496 | 2022 | PMID: 35239701 Open Access |
| Zhang 2012 Nat Protoc | Zhang J, Nuebel E, Wisidagama DR, Setoguchi K, Hong JS, Van Horn CM, Imam SS, Vergnes L, Malone CS, Koehler CM, Teitell MA (2012) Measuring energy metabolism in cultured cells, including human pluripotent stem cells and differentiated cells. https://doi.org/10.1038/nprot.2012.048 | 2012 | Nat Protoc 7: 1068-85. PMID: 22576106 Open Access |
| MiPNet18.10 O2k-Specifications | O2k-specifications for respirometry and comprehensive OXPHOS analysis. | 2024-03-26 | |
| MiPNet06.03 POS-calibration-SOP | O2k Quality Control 1: Polarographic oxygen sensors and accuracy of calibration. | 2023-02-06 | |
| MiPNet19.18B POS-service | Service of the polarographic oxygen sensor OroboPOS. | 2021-06-23 | |
| MiPNet14.06 Instrumental O2 background | O2k Quality Control 2: Instrumental oxygen background correction and accuracy of oxygen flux. | 2023-10-19 | |
| MiPNet19.03 O2k-cleaning and ISS | O2k-Chamber cleaning SOP and Integrated Suction System (ISS). | 2023-03-14 |
Support
- This work was part of the Oroboros NextGen-O2k project, with funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement nº 859770.
Labels: MiParea: Respiration, Instruments;methods
Tissue;cell: HEK
HRR: Oxygraph-2k, O2k-Protocol
Bioblast 2022, SUIT-001 O2 ce-pce D003