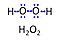From Bioblast
Description
Hydrogen peroxide, H2O2 or dihydrogen dioxide, is one of several reactive oxygen intermediates generally referred to as reactive oxygen species (ROS). It is formed in various enzyme-catalyzed reactions (e.g., superoxide dismutase) with the potential to damage cellular molecules and structures. H2O2 is dismutated by catalase to water and oxygen. H2O2 is produced as a signaling molecule in aerobic metabolism and passes membranes more easily compared to other ROS.
Abbreviation: H2O2
MitoPedia O2k and high-resolution respirometry:
O2k-Open Support
Measurement of hydrogen peroxide
- Two techniques to measure H2O2 concentration and H2O2 production can be combined with the O2k:
- O2k-Fluorometry
- O2k-Amperometric Module - electrochemical measurement with an amperometric H2O2 sensor.
- Two techniques to measure H2O2 concentration and H2O2 production can be combined with the O2k:
Fluorescence
- The fluorometric measurement of H2O2 in the O2k is based on the O2k-Fluo LED2-Module, using Amplex UltraRed (or one of its brand names).[1]
- » Amplex UltraRed
- » Komlodi 2021 MitoFit AmR-O2 — AmplexRed, O2 dependence, yeast
- » MiPNet20.14 AmplexRed H2O2-production
- » MiPNet18.05_Amplex-Mouse-heart
- » MiPNet17.17_Amplex-Mouse-brain
Electrochemical measurement
- For electrochemically (H2O2 electrode) measuring H2O2 (and all other amperometric methods) please see our O2k-NO Amp-Module and its manual MiPNet15.05 NO-manual. All stated there about measuring NO is technically also correct for using a H2O2 electrode together with the oxygraph. In this manual you will also find which accessories you need to use a H2O2 electrode together with the O2k-MultiSensor System (there are different requirements for different generations of O2ks). The electrode itself has to be bought from a third party.
- It is sometimes stated that the available H2O2 electrodes may not have the required sensitivity for most ROS studies. WPI states a lower detection limit of "<100 nM" for their 2 mm "macro" electrode and "<10 nM" for their 0.1 mm "mini sensor". The latter value is quite similar to the stated detection limit for fluorescence based methods, however it is not clear if the "mini sensor", probable developed for direct tissue insertion, works well in macroscopic chambers. A direct comparison would be helpful. Please add your experiences!
- Picomolar detection ranges have been reported for a method combining Amplex Red (R) with square wave voltammetry.[2]
- ↑ Makrecka-Kuka M, Krumschnabel G, Gnaiger E (2015) High-resolution respirometry for simultaneous measurement of oxygen and hydrogen peroxide fluxes in permeabilized cells, tissue homogenate and isolated mitochondria. Biomolecules 5:1319-38. - »Bioblast link«.
- ↑ Lyon JL, Stevenson KJ (2006) Picomolar peroxide detection using a chemically activated redox mediator and square wave voltammetry. Anal Chem 78:8518-25. - »Bioblast link«
Preparation of H2O2 stock solutions
H2O2 calibration
Reoxygenation of the respiration medium with H2O2
- » Refer to MiPNet14.13 for H2O2 preparation.
SUITbrowser questions: H2O2 production
- SUIT protocols can give information on different aspects related to H2O2 production, such as:
- Mt-membrane potential dependence of H2O2 production
- [O2] dependence of H2O2 production
- RET-initiated H2O2 production
- NADH-pathway initiated H2O2 production
- SUIT protocols can give information on different aspects related to H2O2 production, such as:
- With the SUITbrowser it is possible to find the best SUIT protocols to answer these and other research questions.
- Bioblast links: Hydrogen peroxide - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Specific
- General
MitoPedia topics:
Substrate and metabolite
Labels:
MitoPedia:NextGen-O2k