Krajcova 2016 Abstract MitoFit Science Camp 2016
| Mitochondrial pathogenesis of propofol infusion syndrome in an in vitro model of human skeletal muscle. |
Link:
Krajcova A, Waldauf P, Andel M, Duska F (2016)
Event: MitoFit Science Camp 2016 Kuehtai AT
Propofol infusion syndrome is a rare, but serious adverse effect of a commonly used drug with a very high mortality rate (˃50%) [1]. The symptoms can occur in various combinations and include: unexplained metabolic acidosis, arrhythmia, Brugada like pattern on electrocardiograph (elevated ST-segment and coved-T wave), cardiac and/or renal failure, rhabdomyolysis, hyperkalaemia, hepatomegaly and hyperlipidaemia. The mechanism of the syndrome is still unknown: experimental studies performed on animal models and clinical features of the syndrome are suggestive of its mitochondrial origin. We hypothesize that propofol decreases respiratory chain capacity, inhibits fatty acid oxidation and induces inner mitochondrial membrane uncoupling in a dose-dependent manner. Our study aims to test this hypothesis in an in vitro model of human skeletal muscle by acute and chronic exposure of propofol.
Skeletal muscle homogenates were prepared from biopsies obtained from patients (n=16) undergoing hip replacement surgery. Afterwards, energy metabolism after propofol exposure (at concentrations from 25 up to 400 µg/ml) was assessed by Oroboros Oxygraph-2k. Firstly, oxygen consumption rate (OCR) was measured at baseline, after addition of ATPase inhibitor and subsequently after incubation in a range of propofol concentrations (for 3 minutes) to induce its possible uncoupling effect. In following experiments, specific substrates were added to test possible effect of propofol on individual complexes. Fatty acid oxidation was measured as etomoxir-inhibitable OCR after adding of palmitoylcarnitine. Skeletal muscle cells were isolated from biopsies as well and after that exposed to a range of propofol concentrations resembling clinical levels in human plasma during propofol infusion (0, 1, 2.5, 5 a 10 µg/ml) and to its lipid vehicle (Intralipid® - IL).
• O2k-Network Lab: CZ Prague Krajcova A
Labels: MiParea: Respiration, Patients, Pharmacology;toxicology
Organism: Human
Tissue;cell: Skeletal muscle
Preparation: Homogenate
Enzyme: Complex I, Complex II;succinate dehydrogenase, Complex IV;cytochrome c oxidase
Coupling state: LEAK, ET Pathway: F HRR: Oxygraph-2k Event: D2 MitoFit Science Camp 2016
Affiliations and Support
1-Lab Metabolism Bioenergetics, Third Fac Med, Charles Univ Prague; 2-Centre Research Diabetes, Metabolism Nutrition, Third Fac Med, Czech Republic; 3-Dept Anaesthesiology Intensive Care, Third Fac Med, Charles Univ Prague, Czech Republic. - adela.krajcova@seznam.cz
The work was supported by grants GAUK 270915 and PRVOUK P31.
Figure 1
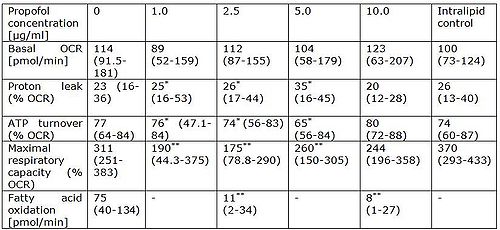
Table 1.
The influence of propofol on mitochondrial metabolism. Data are presented as median (interquartile range). Statistically significant results are signed as * if p-value < 0.05, ** p-value < 0.001.
Abstract continued
After 96 hours of exposure, mitochondrial metabolism was assessed by extracellular flux analysis (Seahorse Biosciences). Oxygen consumption rate (OCR) was measured at baseline and after addition of ATPase inhibitor, mitochondrial uncoupler and complex III inhibitor. Injection of these agents enabled to calculate baseline OCR, ATP turnover rate, proton leak through inner mitochondrial membrane and respiratory chain capacity (uncoupled respiration). Similarly as in tissue homogenates, the capacity of fatty acid oxidation was measured as etomoxir-inhibitable OCR after adding of uncoupler and palmitate. Values presented in Table are expressed as % of baseline OCR.
In human skeletal muscle homogenates, propofol started to inhibit complex I and II at the concentration of 25 µg/ml (with more sensitivity to complex I) after its acute exposure, whilst it did not influence complex IV nor in the highest concentration (at 400 µg/ml). In addition, propofol acts as an uncoupler at concentration of 50 µg/ml and as a partial inhibitor of fatty acid oxidation at the concentration ≥ 100 µg/ml. In human skeletal muscle cells exposed to propofol, effect of prolonged incubation showed that respiratory chain capacity was decreased and uncoupling of inner mitochondrial membrane was increased. The most significant result was propofol-induced inhibition of fatty acid oxidation to 15%, respectively 11% of baseline values. Interestingly, we did not observe a significant inhibition of complex I, II or IV in comparison with acute propofol exposure in tissue homogenates.
References
- Krajcova A, Waldauf P, Andel M, Duska F (2015) Propofol infusion syndrome: a structured review of experimental studies and 153 published case reports. Crit Care 12:398.