From Bioblast
| News and Events | Working Groups | Short-Term Scientific Missions | Management Committee | Members |
COST Action CA15203 (2016-2021): MitoEAGLE
Evolution-Age-Gender-Lifestyle-Environment: mitochondrial fitness mapping
MitoEAGLE data: blood and cultured cells
WG4 Project application
- MitoEAGLE data repository for blood cells and clutured cells
Description
- Biopsy sampling of human muscle and fat tissue for mitochondrial studies is an invasive approach. More recently measurement of mitochondrial function in human blood cells – as well as temporary cryopreservation (Karabatsiakis 2014 Transl Psychiatry) – has become an attractive and by far less invasive alternative for mitochondrial diagnostics. Mitochondrial function of blood cells was linked with numerous diseases and metabolic conditions such as sepsis, neurodegenerative diseases, depression, hypertension, amyotrophic lateral sclerosis, and early-stage heart failure (e.g. Sjoevall 2013 Crit Care; Karabatsiakis 2014 Transl Psychiatry; Hagl 2014 J Alzheimer Dis). The use of human blood cells will thus predictably widen the applicability of respirometry in human physiology and will provide a significant stimulus for the entire field of mitochondrial medicine.
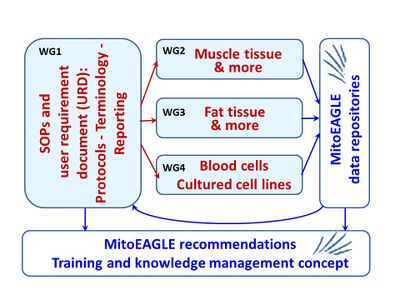
- A key towards achieving comparability among data sets and the applicability of human blood cells for the evaluation of mitochondrial fitness in health and disease is the standardization of the procedures to separate or isolate blood cells, the experimental procedure for the evaluation of mitochondrial respiratory characteristics, and the format for reporting. A consensus on protocols should be reached in WG4, calling for a Workshop right at the start of the COST Action. Depending on the WG4 participants input, cell lines will be defined for which harmonization of datasets should be achieved (Fig. 2).
Management
Tasks
- Kick-off meeting to develop a working plan for the establishment of consensus protocols, reporting schemes, and the assignment of specific study tasks.
- Development of SOPs for cell separation.
- Development of laboratory protocols for individual blood cell types (and other cell types) for mitochondrial studies on intact and permeabilized cells.
- Application of SOPs in experimental studies during Short Term Scientific Missions and feeding of data into the MitoEAGLE data repository.
- Publication of SOPs and study results.
Milestones
- Consensus on protocols, reporting schemes and work assignments.
- Completed SOPs for cell preparation & laboratory protocols.
- Application study finished and data transmitted to MitoEAGLE data repository.
- Publication finished.
Deliverables
- SOPs for blood cell separation and respirometric characterisation open for the research community.
- MitoEAGLE data repository for comparative data evaluation, planning of future studies, data mining.
- Publication with a set of reference data.