Serna 2022 Abstract Bioblast
| 5.2. «10+5» Serna Julian DC, de Miranda Ramos V, Cabral-Costa JV, Vilas-Boas EA, Amaral AG, Ohya G, Caldeira da Silva CC, Kowaltowski AJ (2022) Measuring mitochondrial Ca2+ efflux in isolated mitochondria and permeabilized cells. Bioblast 2022: BEC Inaugural Conference. In: https://doi.org/10.26124/bec:2022-0001 »Bioenergetics Communications« |
Link: Bioblast 2022: BEC Inaugural Conference
Serna Julian DC, de Miranda Ramos Vitor, Cabral-Costa Joao Victor, Vilas-Boas Eloisa A, Amaral Andressa G, Ohya Georgia, Caldeira da Silva Camille C, Kowaltowski Alicia J (2022)
Event: Bioblast 2022
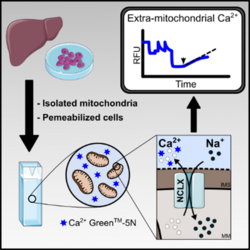
Mitochondrial Ca2+ efflux is essential for mitochondrial and cell Ca2+ homeostasis. Mitochondrial inner membrane Ca2+/H+ and Na+/Li+/Ca2+ (NCLX) exchangers are known today to be plastic transporters [1], with important roles in physiological responses and pathological states. Until now, however, no consensus protocols were available to measure mitochondrial Ca2+ efflux, and we find that some published protocols may induce mitochondrial permeability transition pore (mtPTP) opening, underestimating the effects of these exchangers. In this work we describe a method to measure Na+-sensitive and insensitive mitochondrial Ca2+ efflux activity in isolated mitochondria and permeabilized cells.
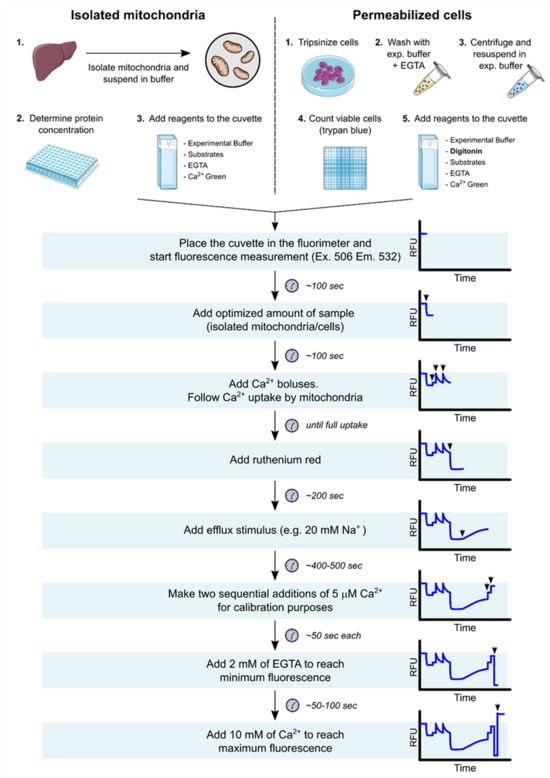
Mitochondria were isolated from mouse liver and rat heart. PLC/PRF/5 (hepatoma) and INS-1E (insulinoma) cells were permeabilized with digitonin. Digitonin concentrations were titrated using a high-resolution respirometer (OROBOROS). Outer mitochondrial membrane (OMM) integrity was assessed with exogenous cytochrome c. Calcium transport was measured following concentrations in the extramitochondrial medium using Ca2+-Green 5N and an Hitashi F4500 Fluorimeter. The efflux protocol was performed as follows (figure 1): Mitochondria (isolated or in permeabilized cells), were loaded with a non-permeability transition inducing amount of Ca2+ and treated with ruthenium red to inhibit uptake. Ca2+ efflux was measured under non-stimulated conditions and also stimulated by 20 mM NaCl or LiCl. Ca2+-Green 5N Kd values were determined. Fluorescence values were always transformed in [Ca2+] for quantifications.
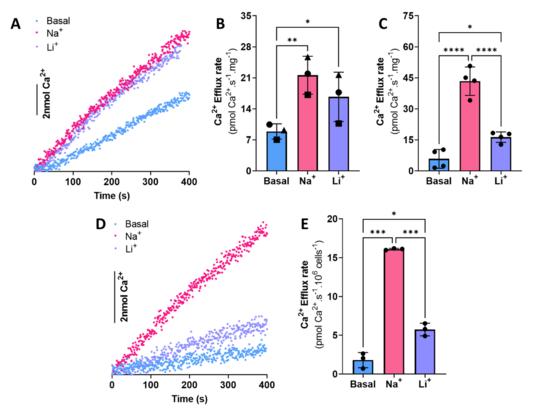
Widespread mtPTP formation is easily identifiable when measuring Ca2+ fluxes, as it leads to overt Ca2+ release, but we find it can often be overlooked when affecting a subset of the mitochondrial population. Total efflux rates increase with increasing Ca2+ loads due to a CsA-sensitive activity, which indicates that the mtPTP was responsible [2]. Our results demonstrate that using higher Ca2+ loads promote mtPTP opening even when the Ca2+ retention capacity has not been exceeded. Liver NCLX-mediated Ca2+ efflux activity had not been detected in many prior work [3,4]. In mice liver mitochondria, we were able to measure both Na+- and Li+-stimulated Ca2+ efflux, and to distinguish it from Ca2+/H+ exchange. Our protocol produces similar results in isolated rat heart mitochondria. Interestingly, NCLX activity was predominant in heart relative to Ca2+/H+ exchange. In digitonin-permeabilized PCL and INS-1E cells, we were also able to measure/characterize NCLX activity and to distinguish it from Ca2+/H+ exchange. The last approach provides an alternative path to measure mitochondrial efflux in cells when microscopy is not available, also leaving behind the restrains (and the complexity) present in intact cell models.
Overall, our approach allows us to dissect between Na+-sensitive and insensitive Ca2+ efflux. We demonstrate that a vital point in obtaining consistent and reliable Ca2+ extrusion activity measurements through mitochondrial exchangers is to avoid mtPTP opening by either using low Ca2+ loads or adding cyclosporin A to all traces. In the absence of this step, at least part of the activity of NCLX and Ca2+/H+ exchange may be masked by mtPTP-promoted permeabilization. Using this method, we were able to demonstrate NCLX activity in mouse liver mitochondria and permeabilized liver hepatoma PLC/PRF/5 cells. Additionally, we validated our method in isolated rat heart mitochondria, as well as the insulinoma cell line INS-1E.
- Giorgi C, Marchi S, Pinton P (2018) The machineries, regulation and cellular functions of mitochondrial calcium. https://doi.org/10.1038/s41580-018-0052-8
- Vercesi AE, Castilho RF, Kowaltowski AJ, de Oliveira HCF, de Souza-Pinto NC, Figueira TR, Busanello ENB (2018) Mitochondrial calcium transport and the redox nature of the calcium-induced membrane permeability transition. https://doi.org/10.1016/j.freeradbiomed.2018.08.034
- Haworth RA, Hunter DR, Berkoff HA (1980) Na+ releases Ca2+ from liver, kidney and lung mitochondria. https://doi.org/10.1016/0014-5793(80)80076-7
- Rysted JE, Lin Z, Walters GC, Rauckhorst AJ, Noterman M, Liu G, Taylor EB, Strack S, Usachev YM (2021) Distinct properties of Ca2+ efflux from brain, heart and liver mitochondria: The effects of Na+, Li+ and the mitochondrial Na+/Ca2+ exchange inhibitor CGP37157. https://doi.org/10.1016/j.ceca.2021.102382
• Keywords: NCLX, mtPT, Isolated mitochondria, Permeabilized cells, Calcium
• O2k-Network Lab: BR Sao Paulo Kowaltowski AJ
Affiliations
- Serna JDC1, de Miranda Ramos V1, Cabral-Costa JV1, Vilas-Boas EA1, Amaral AG2, Ohya G1, Caldeira da Silva CC1, Kowaltowski AJ1
- Departamento de Bioquímica, Instituto de Química, Universidade de São Paulo, 05508-900, Brazil
- Departamento de Fisiologia, Instituto de Ciências Biomédicas, Universidade de São Paulo, 05509-900, Brazil
Figures
List of abbreviations, terms and definitions - MitoPedia
Labels:
Event: B1