Acuna-Castroviejo 2018 MiP2018
| Mitochondria, melatonin, and neuroinflammation in Parkinson's disease. Acuna-Castroviejo_Presentation |
Link: MiP2018
Acuna-Castroviejo D, Lopez A, Diaz-Casado ME, Fernandez-Ortiz M, Fernandez-Martinez J, Escames G (2018)
Event: MiP2018
Melatonin is synthetized by the mitochondria, which in turn are the main intracellular targets of the indoleamine [1,2]. Due to the impairment of mitochondria in Parkinson's disease (PD), and its relationship with neuroinflammation, we analyzed the mitochondrial bioenergetics and melatonin effects in two models of parkinsonism, mouse and zebrafish. The participation of inflammation on mitochondria disfunction was analyzed in iNOS and nNOS deficient mice. Mitochondria were prepared from substantia nigra and striatum of control and MPTP-treated mice, and bioenergetics analyzed in an Oxygraph-2K respirometer [3]. Our results showed that MPTP increased iNOS activity in substantia nigra and striatum, whereas it sharply reduced complex I activity and mitochondrial bioenergetics in all strains. In the presence of MPTP, mice lacking iNOS showed similar restricted mitochondrial function than wild type or mice lacking nNOS. Therefore, neuroinflammation and mitochondrial dysregulation seem to act in parallel in the MPTP model of PD. Melatonin administration counteracted these effects, preventing from the drastic changes in mitochondrial oxygen consumption, and neuroinflammation, recovering normal locomotor activity of mice. The protective effects of melatonin on mitochondria are also independent of its anti-inflammatory properties, but both effects are required for an effective anti-parkinsonian activity of the indoleamine as reported in this study.
In the second study, we studied the capacity of melatonin to recover from a parkinsonian phenotype [4]. Here, 24 to 72 hpf zebrafish were treated with MPTP and in vivo respiration was measured in a Seahorse respirometer. A reduction in electron transfer system capacity, ATP turnover, and increased proton leak, were observed at 72 hpf in MPTP-treated embryos. These changes were followed by neuroinflammation and autophagy impairment. After removing MPTP from the treatment at 72 hpf, these bioenergetic deficiencies persisted up to 120 hpf. The administration of melatonin to zebrafish embryos at 72 hpf, when mitochondrial dysfunction is already present, restored the respiratory capacity and ATP production, reduced neuroinflammation, and normalized autophagy. Melatonin, added together with MPTP or added once MPTP was removed, prevented and recovered, respectively, the parkinsonian phenotype once it was established, restoring gene expression and normal function of the parkin/PINK1/DJ-1/MUL1 loop and also the normal motor activity of the embryos.
• Bioblast editor: Plangger M, Kandolf G
• O2k-Network Lab: ES Granada Acuna-Castroviejo D
Labels: MiParea: Respiration Pathology: Parkinson's
Organism: Mouse Tissue;cell: Nervous system
Coupling state: LEAK, OXPHOS Pathway: N HRR: Oxygraph-2k
Affiliations
Acuña-Castroviejo D(1,2), López A(1,3), Díaz-Casado ME()1, Fernández-Ortiz M(1), Fernández-Martínez J(1), Escames G(1,2)
- Dept Fisiología, Fac Medicina, Centro Investigación Biomédica, Parque Tecnológico Ciencias de la Salud, Univ Granada
- CIBERfes, Ibs.Granada, UGC de Laboratorios Clínicos, Complejo Hospitalario Granada; Spain. - dacuna@ugr.es
- Dept Medical Genetics, Univ Cambridge, Cambridge Inst Medical Research, Addenbrooke's Hospital, Hills Road, Cambridge, UK.
Figures
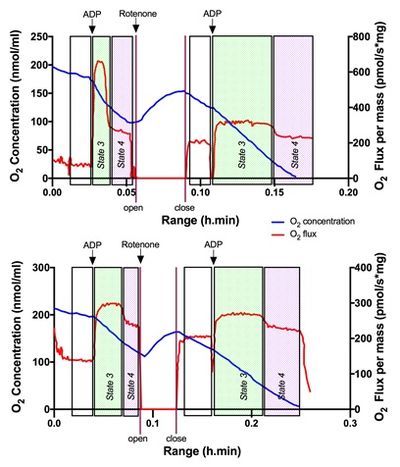
Figure 1. Oxygraph trace of mitochondrial respiration in SN control (above) and MPTP (down) from wild-type for iNOS mice. Oxygen concentration (nmol/mL) and oxygen flux per mass (pmol/s*mg protein) are represented. Black arrows represent activation of respiration by additions of ADP (400 nmol) before and after complex I inhibition by the addition of 2.5 μM of rotenone (white arrow). Changes in state 3 and 4 were then evaluated.
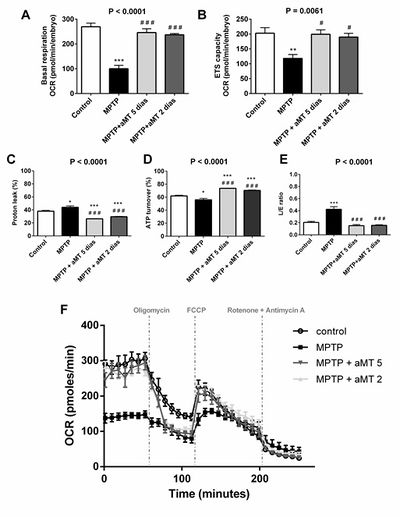
Figure 2. Melatonin repaired mitochondrial impairment in 120 hpf embryos. (A) Basal respiration. (B) ETS capacity. (C) Percentage of proton leak. (D) L/E (proton leak/ETS) ratio. Melatonin restored normal mitochondrial bioenergetics; (E) ATP turnover. (F) representative recordings of the OCR obtained by respiratory analysis in zebrafish embryos in vivo. Data expressed as described in Figure 1. *p < 0.05, **p < 0.01, and ***p < 0.001 versus control; #p < 0.05, and ###p < 0.001 versus MPTP.
References and Support
- Suofu Y, Li W, Jean-Alphonse FG, Jia J, Khattar NK, Li J, Baranov SV, Leronni D, Mihalik AC, He Y, Cecon E, Wehbi VL, Kim J, Heath BE, Baranova OV, Wang X, Gable MJ, Kretz ES, Di Benedetto G, Lezon TR, Ferrando LM, Larkin TM, Sullivan M, Yablonska S, Wang J, Minnigh MB, Guillaumet G, Suzenet F, Richardson RM, Poloyac SM, Stolz DB, Jockers R, Witt-Enderby PA, Carlisle DL, Vilardaga JP, Friedlander RM (2017) Dual role of mitochondria in producing melatonin and driving GPCR signaling to block cytochrome c release. PNAS doi/10.1073/pnas.1705768114.
- Acuña-Castroviejo D, López LC, Escames G, López A, García JA, Reiter RJ (2011) Melatonin-mitochondria interplay in health and disease. Curr Top Med Chem 11:221-40.
- López A, Ortiz F, Doerrier C, Venegas C, Fernández-Ortiz M, Aranda P, et al (2017) Mitochondrial impairment and melatonin protection in parkinsonian mice do not depend of inducible or neuronal nitric oxide synthases. Plos One doi:10.1371/journal.pone.0183090.
- Díaz-Casado ME, Rusanova I, Aranda P, Fernández-Ortiz M, Sayed RKA, Fernández-Gil BI, Hidalgo-Gutiérrez A, Escames G, López LC, Acuña-Castroviejo D (2017) In vivo determination of mitochondrial respiration in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated zebrafish reveals the efficacy of melatonin in restoring mitochondrial normalcy. Zebrafish 15:15-26.
Supported by grants P10-CTS-5784 (Junta de Andalucía, Spain); PI13-00981, RD12/0043/0005, and CB16-10-00238 (Ministerio de Competitividad and Fondos Feder, Spain).