Description
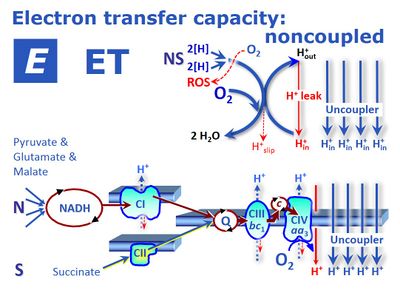
![]() ETS capacity is the respiratory electron transfer system capacity, E, of mitochondria in the experimentally induced noncoupled state. The conditions for measurement and expression of respiration vary (oxygen flux in state E, JO2E or oxygen flow in state E, IO2E). If these conditions are defined and remain consistent within a given context, then the simple symbol E for respiratory state is used to substitute the more explicit expression for respiratory activity. In state E, the mt-membrane potential is almost fully collapsed and provides a reference state for flux control ratios. In intact mitochondria, the ETS capacity depends not only on the inner membrane-bound ETS (mETS, with respiratory Complexes CI to CIV, electron-transferring flavoprotein ETF, and glycerophosphate dehydrogenase) but also integrates transporters across the inner mt-membrane, the TCA cycle and other matrix dehydrogenases. Its experimental determination in mitochondrial preparations or intact cells requires the measurement of oxygen consumption in the presence of defined substrates and of an established uncoupler at optimum concentration. This optimum concentration is determined by stepwise titration of the uncoupler up to the concentration inducing maximum flux.
» MiPNet article
ETS capacity is the respiratory electron transfer system capacity, E, of mitochondria in the experimentally induced noncoupled state. The conditions for measurement and expression of respiration vary (oxygen flux in state E, JO2E or oxygen flow in state E, IO2E). If these conditions are defined and remain consistent within a given context, then the simple symbol E for respiratory state is used to substitute the more explicit expression for respiratory activity. In state E, the mt-membrane potential is almost fully collapsed and provides a reference state for flux control ratios. In intact mitochondria, the ETS capacity depends not only on the inner membrane-bound ETS (mETS, with respiratory Complexes CI to CIV, electron-transferring flavoprotein ETF, and glycerophosphate dehydrogenase) but also integrates transporters across the inner mt-membrane, the TCA cycle and other matrix dehydrogenases. Its experimental determination in mitochondrial preparations or intact cells requires the measurement of oxygen consumption in the presence of defined substrates and of an established uncoupler at optimum concentration. This optimum concentration is determined by stepwise titration of the uncoupler up to the concentration inducing maximum flux.
» MiPNet article
Abbreviation: E
Reference: Gnaiger 2014 MitoPathways, Gnaiger 2009 Int J Biochem Cell Biol
MitoPedia methods:
Respirometry
MitoPedia topics: "Respiratory state" is not in the list (Enzyme, Medium, Inhibitor, Substrate and metabolite, Uncoupler, Sample preparation, Permeabilization agent, EAGLE, MitoGlobal Organizations, MitoGlobal Centres, ...) of allowed values for the "MitoPedia topic" property.
Respiratory state"Respiratory state" is not in the list (Enzyme, Medium, Inhibitor, Substrate and metabolite, Uncoupler, Sample preparation, Permeabilization agent, EAGLE, MitoGlobal Organizations, MitoGlobal Centres, ...) of allowed values for the "MitoPedia topic" property.
Why ETS, why not State 3u?
| Gnaiger E (2014) Why ETS, why not State 3u? Mitochondr Physiol Network 2014-07-06. |
Abstract: ![]() Measurement of ETS capacity in the noncoupled state at optimum uncoupler concentration does not represent a general substitute for determination of OXPHOS capacity (compare State 3). If the ratio of OXPHOS/ETS capacity (P/E ratio) is less than one, noncoupled respiration overestimates the apparent reserve capacity for oxidative phosphorylation with respect to ROUTINE respiration of intact cells.
Measurement of ETS capacity in the noncoupled state at optimum uncoupler concentration does not represent a general substitute for determination of OXPHOS capacity (compare State 3). If the ratio of OXPHOS/ETS capacity (P/E ratio) is less than one, noncoupled respiration overestimates the apparent reserve capacity for oxidative phosphorylation with respect to ROUTINE respiration of intact cells.
• O2k-Network Lab: AT Innsbruck Gnaiger E
Labels:
Coupling state: ETS"ETS" is not in the list (LEAK, ROUTINE, OXPHOS, ET) of allowed values for the "Coupling states" property.
HRR: Theory
Electron transfer system versus electron transport chain
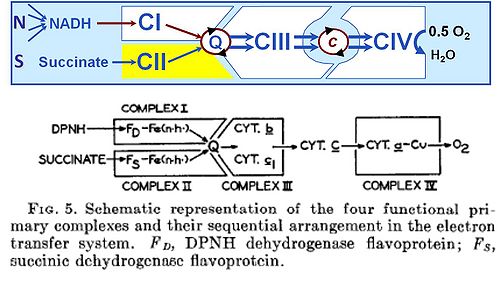
The well established terms 'respiratory chain' or 'electron transfer chain' suggest erroneously that the convergent electron transfer system may be designed as a simple chain. But the term electron transport chain (or electron transfer chain, ETC) is a misnomer. Understanding mitochondrial respiratory control has suffered greatly from this inappropriate terminology, although textbooks using the term ETC (Lehninger 1970) make it sufficiently clear that electron transfer systems are not arranged as a chain: the „ETC‟ is in fact not a simple chain but an arrangement of electron transfer complexes in a non-linear, convergent electron transfer system. The classically introduced term electron transfer system (Hatefi et al 1962 [1]) is accurate and sufficient. [2]
The established convention of defining the 'electron transport chain' as being comprised of four Complexes has conceptual weaknesses.
(a) In fact, there are at least six Complexes of mitochondrial electron transfer: In addition to Complexes I and II, glycerophosphate dehydrogenase complex (CGpDH) and electron-transferring flavoprotein complex (CETF) are involved in the Q-junction with electron transfer to Complex III. [3],[4]
(b) The term „chain‟ suggests a linear sequence, whereas the functional structure of the electron transfer system can only be understood by recognizing the convergence of electron flow at the Q-junction, followed by a chain of Complexes III and IV, mediated by cytochrome c. [5]
Electrons flow to oxygen from either Complex I with a total of three coupling sites, or from Complex II and other flavoproteins, providing multiple entries into the Q-cycle with two coupling sites downstream. [6]
Electron transfer versus transport
Electron transfer and electron transport are used synonymously. A general distinction, however, may be helpful:
(i) Transfer (inter- or intramolecular) of a reactant involves a chemical reaction.
(ii) Transport (from one place to another) of an entity is a (vectorial) process in contrast to a chemical reaction. [7]
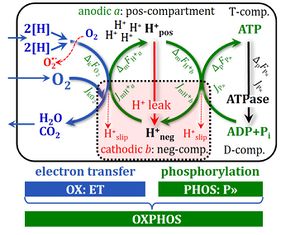
The important difference between states P and E
The abbreviation State 3u is used frequently in bioenergetics, to indicate the noncoupled state of maximum respiration, E,[8] without sufficient emphasis on the fundamental difference between state P (OXPHOS capacity; coupled, with an uncoupled component; State 3) and state E (ETS capacity, noncoupled).[9],[10]
- P=E: The specific case of equal OXPHOS and ETS capacity (P/E=1) yields the important information that the capacity of the phosphorylation system matches or is in potential excess of the ETS capacity, such that OXPHOS capacity is not limited by the phosphorylation system in the specific mitochondria. This varies with species and tissues, and changes as a result of pathologies due to defects in the phosphorylation system. An example for P/E=1 is mouse skeletal muscle mitochondria. [11]
- P<E: When OXPHOS is less than ETS capacity, the phosphorylation system limits OXPHOS capacity, and there is an apparent ETS excess capacity. For example, this is the case in healthy human skeletal muscle mitochondria. [12]
- P>E: If ETS is less than OXPHOS capacity in intact cells, or in mitochondrial preparations with defined substrate(s), then you have encountered an experimental artefact, and the apparent ETS capacity is too low. Artificially low ETS capacity may be obtained due to overtitration of uncoupler. Inhibitors of ATP synthase may suppress ETS capacity in intact cells, particularly in stressed cells.
Consequences for evaluation of coupling
In some textbooks on Bioenergetics, the RCR is defined as either the State 3/State 4 ratio or the State 3u/State 4 ratio. This reflects lack of conceptual distinction between State 3 (or P) and 3u (E), and clarification is best achieved by avoiding ambiguous terminology. RCR as defined originally is the 'acceptor control ratio' or 'adenylate control ratio' (see LEAK control ratio, L/E).[13] ETS capacity but not OXPHOS capacity provides a valid reference for an index of uncoupling.
Related terms in Bioblast
![]() OXPHOS, P
OXPHOS, P
![]() ROUTINE, R
ROUTINE, R
![]() ETS, E
ETS, E
![]() LEAK, L
LEAK, L
 ROX, R
ROX, R
The ETS state
- ETS-competent substrate state
- Level flow
- Noncoupled respiration - Uncoupler[14]
- Coupling control protocol
- Biochemical coupling efficiency[15]
- E-L coupling control factor - Excess E-P capacity factor - Excess E-R capacity factor
- Coupling control ratio - Uncoupling control ratio
- LEAK control ratio - OXPHOS control ratio
- ROUTINE control ratio - NetROUTINE control ratio
References
- ↑ Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer system XLII. Reconstitution of the electron transfer system. J Biol Chem 237:2661-9. »Open Access«
- ↑ International Union of Biochemistry (1991) Nomenclature of electron-transfer proteins. Biochim Biophys Acta 1060. »Open Access«
- ↑ International Union of Biochemistry (1991) Nomenclature of electron-transfer proteins. Biochim Biophys Acta 1060. »Open Access«
- ↑ Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. OROBOROS MiPNet Publications, Innsbruck:80 pp. »Open Access«
- ↑ Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. OROBOROS MiPNet Publications, Innsbruck:80 pp. »Open Access«
- ↑ Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. OROBOROS MiPNet Publications, Innsbruck:80 pp. »Open Access«
- ↑ International Union of Biochemistry and Molecular Biology. Recommendations for terminology and databases for biochemical thermodynamics - The IUPAC Green Book. »Open Access«
- ↑ Gnaiger E. Electron transfer system versus electron transport chain. Mitochondr Physiol Network. »Electron transfer system«
- ↑ Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. »PMID: 19467914«
- ↑ Gnaiger E (2014) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 4th ed. Mitochondr Physiol Network 19.12. OROBOROS MiPNet Publications, Innsbruck:80 pp. »Open Access«
- ↑ Aragonés J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, Dirkx R, Zacchigna S, Lemieux H, Jeoung NH, Lambrechts D, Bishop T, Lafuste P, Diez-Juan A, K Harten S, Van Noten P, De Bock K, Willam C, Tjwa M, Grosfeld A, Navet R, Moons L, Vandendriessche T, Deroose C, Wijeyekoon B, Nuyts J, Jordan B, Silasi-Mansat R, Lupu F, Dewerchin M, Pugh C, Salmon P, Mortelmans L, Gallez B, Gorus F, Buyse J, Sluse F, Harris RA, Gnaiger E, Hespel P, Van Hecke P, Schuit F, Van Veldhoven P, Ratcliffe P, Baes M, Maxwell P, Carmeliet P (2008) Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. Nat Genet 40:170-80. »Open Access«
- ↑ Pesta D, Hoppel F, Macek C, Messner H, Faulhaber M, Kobel C, Parson W, Burtscher M, Schocke M, Gnaiger E (2011) Similar qualitative and quantitative changes of mitochondrial respiration following strength and endurance training in normoxia and hypoxia in sedentary humans. Am J Physiol Regul Integr Comp Physiol 301:R1078–87. »Open Access«
- ↑ Gnaiger E. Biochemical coupling efficiency: from 0 to <1. Mitochondr Physiol Network. »Biochemical coupling efficiency«
- ↑ Gnaiger E. Is respiration uncoupled - noncoupled - dyscoupled? Mitochondr Physiol Network. »Uncoupler«
- ↑ Gnaiger E. Biochemical coupling efficiency: from 0 to <1. Mitochondr Physiol Network. »Biochemical coupling efficiency«
- Bioblast alert 2014(04)
- Bioblast alert 2013(02): CCCP versus FCCP for high-resultion respirometry (HRR).