Chance 1955 J Biol Chem-I
| Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. J Biol Chem 217:383-93. |
Chance B, Williams GR (1955) J Biol Chem
Abstract:
The abrupt initiation and cessation of rapid respiration of “tightly coupled” oxidative phosphorylation systems caused by the addition and the utilization of a known concentration of phosphate acceptor can be satisfactorily measured by a rapidly responding polarographic technique utilizing a vibrating platinum micro electrode. The ratios of added ADP to oxygen utilized (ADP: 0) are 3.1, 2.6, and 1.8 for glutamate, β-hydroxybutyrate, and succinate, respectively. These values are within the range of P:O ratios obtained by Copenhaver and Lardy (21). The errors in the measurement of ADP and oxygen concentrations are about 5 per cent when about 0.1 µmole of ADP is used. Since negligible ADP is found to be utilized in reactions other than oxidative phosphorylation with these preparations, the ADP:0 values correspond to P:0 ratios.
The initiation of increased respiration of mitochondria following ADP addition requires less than 1 second and is rapid enough to explain measured responses of respiration to physiological activity. The abrupt return of respiration to the initial low level as the added ADP is utilized suggests high affinity for ADP. Half maximal respiration requires only 20 to 30 µM ADP under our experimental conditions.
Selected quotes
- In the course of spectrophotometric studies of the kinetics and nature of the respiratory enzyme systems involved in oxidative phosphorylation (1) it has been essential to assay simultaneously the changes of respiratory rate caused by the presence and absence of phosphate acceptor. A vibrating platinum electrode inserted in the optical cuvette exhibits the necessary accuracy and speed of response. It is found that the initiation and cessation of rapid respiration due to the oxidative phosphorylation of added phosphate acceptor are abrupt enough to mark accurately the amount of oxygen uptake in times as short as 10 to 20 seconds. Thus phosphorylative activity can be directly calculated as the quotient of added ADP to the amount of rapid oxygen utilization. Typical values for the ADP:O ratio are given for various substrates and possible errors and limitations of the method are discussed.
- O2 backdiffusion: The vibration of the electrode disturbs the surface of the liquid and mixes the solution in contact with the air with the bulk of the solution. .. This rate of mixing is about 0.1 µM 02 per second when half of the dissolved oxygen has been used up and increases as the difference of oxygen concentration in air and solution increases.
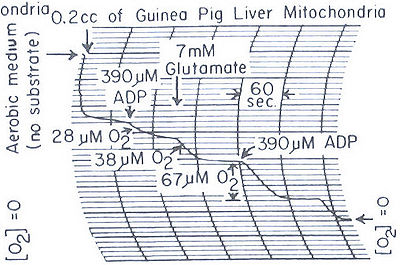
- Endogenous Substrate—If ADP is added to mitochondria in the absence of added substrate, there is considerable oxygen uptake, which in the case of Fig. 4, B amounts to 28 µM oxygen. Addition of glutamate at this point causes a further 38 µM oxygen uptake, which terminates when the ADP is all utilized. The oxygen uptake following ADP addition is presumably caused by a limited amount of endogenous substrate of the mitochondria. The nature of the substrate is not known, but its presence insures that little ADP is present in the mitochondria. The respiration is inhibited by malonate.
- P»/O ratio: In view of the abrupt initiation and cessation of rapid respiration during the utilization of a known ADP concentration, we provide a simple, direct, and rapid means for computing the ADP:O value.
- Specification: The oxygen data are accurate to about 5 per cent in amount and 10 per cent in rate.
- In common with other workers in this field, we do not subtract the respiration rate without phosphate acceptor from that measured during the utilization of added ADP.
Cited by
- Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. https://doi.org/10.26124/bec:2020-0002
- Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. doi:10.26124/bec:2020-0001.v1.
- Cardoso et al (2021) Magnesium Green for fluorometric measurement of ATP production does not interfere with mitochondrial respiration. Bioenerg Commun 2021.1. doi:10.26124/bec:2021-0001
Labels: MiParea: Respiration
Organism: Rat, Guinea pig
Tissue;cell: Liver
Preparation: Isolated mitochondria
Coupling state: LEAK, OXPHOS
Pathway: N
Made history, Respiratory states, LEAK respiration, BEC 2020.1, BEC 2020.2, MitoFit 2021 MgG