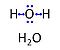Difference between revisions of "Water"
(Created page with "{{MitoPedia |abbr=H<sub>2</sub>O |description=Water, H<sub>2</sub>O, is widely used in the laboratory, particularly as a solvent and cleaning agent. Chemically pure water is used...") |
|||
| Line 1: | Line 1: | ||
{{MitoPedia | {{MitoPedia | ||
|abbr=H<sub>2</sub>O | |abbr=H<sub>2</sub>O | ||
|description=Water, H<sub>2</sub>O, is widely used in the laboratory, particularly as a solvent and cleaning agent. Chemically pure water is used in various grades of purification: double distilled water (ddH<sub>2</sub>O) versus distilled water (AD or dH<sub>2</sub>O) and deionized or demineralized water (diH<sub>2</sub>O). Combination purification methods are frequently used. In published protocols, it is frequently assumed that the standards of each laboratory are applied as to the quality of purified water, when H<sub>2</sub>O is used without further specification. Purification is not only to be controlled with respect to salt content and corresponding electrical conductivity (ultra-pure water: 5.5 μS/m due to H<sup>+</sup> and OH<sup>-</sup> ions), but also in terms of microbial contamination. | |description=[[File:H2O.jpg|left|60px|Water]] | ||
'''Water''', H<sub>2</sub>O, is widely used in the laboratory, particularly as a solvent and cleaning agent. Chemically pure water is used in various grades of purification: double distilled water (ddH<sub>2</sub>O) versus distilled water (AD or dH<sub>2</sub>O) and deionized or demineralized water (diH<sub>2</sub>O). Combination purification methods are frequently used. In published protocols, it is frequently assumed that the standards of each laboratory are applied as to the quality of purified water, when H<sub>2</sub>O is used without further specification. Purification is not only to be controlled with respect to salt content and corresponding electrical conductivity (ultra-pure water: 5.5 μS/m due to H<sup>+</sup> and OH<sup>-</sup> ions), but also in terms of microbial contamination. | |||
}} | }} | ||
{{MitoPedia methods}} | {{MitoPedia methods}} | ||
Revision as of 12:09, 10 July 2014
Description
Water, H2O, is widely used in the laboratory, particularly as a solvent and cleaning agent. Chemically pure water is used in various grades of purification: double distilled water (ddH2O) versus distilled water (AD or dH2O) and deionized or demineralized water (diH2O). Combination purification methods are frequently used. In published protocols, it is frequently assumed that the standards of each laboratory are applied as to the quality of purified water, when H2O is used without further specification. Purification is not only to be controlled with respect to salt content and corresponding electrical conductivity (ultra-pure water: 5.5 μS/m due to H+ and OH- ions), but also in terms of microbial contamination.
Abbreviation: H2O
MitoPedia topics: Media for respirometry, Substrate and metabolite