Lemieux 2011 Int J Biochem Cell Biol
» [[Has info::PMID: 21871578]]
Was written by::Lemieux H, Was written by::Semsroth S, Was written by::Antretter H, Was written by::Hoefer D, Was written by::Gnaiger E (Was published in year::2011) Was published in journal::Int J Biochem Cell Biol
Abstract: [[has abstract:: O2k-in brief
O2k-in brief
Heart failure is a consequence of progressive deterioration of cardiac performance. Little is known about the role of impaired oxidative phosphorylation in the progression of the disease, since previous studies of mitochondrial injuries are restricted to end-stage chronic heart failure. The present study aimed at evaluating the involvement of mitochondrial dysfunction in the development of human heart failure. We measured the control of oxidative phosphorylation with high-resolution respirometry in permeabilized myocardial fibres from donor hearts (controls), and patients with no or mild heart failure but presenting with heart disease, or chronic heart failure due to dilated or ischemic cardiomyopathy. The capacity of the phosphorylation system exerted a strong limitation on oxidative phosphorylation in the human heart, estimated at 121 pmol O2•s˗1•mg˗1 in the healthy left ventricle. In heart disease, a specific defect of the phosphorylation system, Complex I-linked respiration, and mass-specific fatty acid oxidation were identified. These early defects were also significant in chronic heart failure, where the capacities of the oxidative phosphorylation and Electron transfer-pathway per cardiac tissue mass were decreased with all tested substrate combinations, suggesting a decline of mitochondrial density. Oxidative phosphorylation and Electron transfer-pathway capacities were higher in ventricles compared to atria, but the impaired mitochondrial quality was identical in the four cardiac chambers of chronic heart failure patients. Coupling was preserved in heart disease and chronic heart failure, in contrast to the mitochondrial dysfunction observed after prolonged cold storage of cardiac tissue. Mitochondrial defects in the phosphorylation system, Complex I respiration and mass-specific fatty acid oxidation occurred early in the development of heart failure. Targeting these mitochondrial injuries with metabolic therapy may offer a promising approach to delay the progression of heart disease.]] • Keywords: has publicationkeywords::Oxidative phosphorylation, has publicationkeywords::Mitochondrial respiratory control, has publicationkeywords::Human heart disease and heart failure, has publicationkeywords::Permeabilized cardiac fibres, has publicationkeywords::Cold storage, has publicationkeywords::High-resolution respirometry
• O2k-Network Lab: Was published by MiPNetLab::AT Innsbruck Gnaiger E, Was published by MiPNetLab::AT Innsbruck Oroboros, Was published by MiPNetLab::CA Edmonton Lemieux H
Labels: MiParea: MiP area::Respiration, MiP area::mt-Biogenesis;mt-density, MiP area::mt-Medicine, MiP area::Patients
Pathology: Diseases::Cardiovascular
Stress:Injury and adaptation::Ischemia-reperfusion
Organism: Organism::Human
Tissue;cell: tissue and cell::Heart
Preparation: Preparation::Permeabilized tissue
Enzyme: Enzyme::Marker enzyme
Regulation: Topic::Coupling efficiency;uncoupling, Topic::Cyt c, Topic::Fatty acid
Coupling state: Coupling states::LEAK, Coupling states::OXPHOS, Coupling states::ET
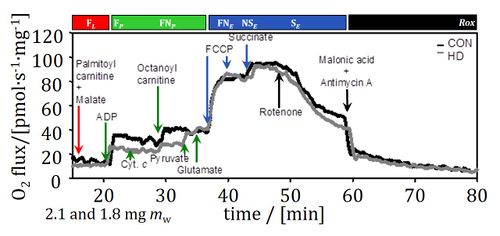
Pathway: Pathways::F, Pathways::N, Pathways::S, Pathways::CIV, Pathways::NS, Pathways::ROX
HRR: Instrument and method::Oxygraph-2k
additional label::1PM;2D;3G;4U;5S;6Rot-, additional label::1PalM;2D;3Oct;4P;5G;6U;7S;8Rot-
SUIT protocols
O2k-Publications
- Increased OXPHOS capacity after cold preservation of the human heart – a paradox resolved by a dyscoupling mechanism.
- Human heart failure: mtDNA versus CS and PGC-1a pathway - PGC-1alpha
- Tissue storage