Sharpley 2006 J Biol Chem
| Sharpley MS, Hirst J (2006) The inhibition of mitochondrial complex I (NADH:ubiquinone oxidoreductase) by Zn2+. J Biol Chem 281:34803-9. https://doi.org/10.1074/jbc.M607389200 |
Sharpley MS, Hirst J (2006) J Biol Chem
Abstract: NADH:ubiquinone oxidoreductase (complex I) from bovine heart mitochondria is a highly complicated, membrane-bound enzyme. It is central to energy transduction, an important source of cellular reactive oxygen species, and its dysfunction is implicated in neurodegenerative and muscular diseases and in aging. Here, we describe the effects of Zn2+ on complex I to define whether complex I may contribute to mediating the pathological effects of zinc in states such as ischemia and to determine how Zn2+ can be used to probe the mechanism of complex I. Zn2+ inhibits complex I more strongly than Mg2+, Ca2+, Ba2+, and Mn2+ to Cu2+ or Cd2+. It does not inhibit NADH oxidation or intramolecular electron transfer, so it probably inhibits either proton transfer to bound quinone or proton translocation. Thus, zinc represents a new class of complex I inhibitor clearly distinct from the many ubiquinone site inhibitors. No evidence for increased superoxide production by zinc-inhibited complex I was detected. Zinc binding to complex I is mechanistically complicated. During catalysis, zinc binds slowly and progressively, but it binds rapidly and tightly to the resting state(s) of the enzyme. Reactivation of the inhibited enzyme upon the addition of EDTA is slow, and inhibition is only partially reversible. The IC50 value for the Zn2+ inhibition of complex I is high (10-50 microm, depending on the enzyme state); therefore, complex I is unlikely to be a major site for zinc inhibition of the electron transport chain. However, the slow response of complex I to a change in Zn2+ concentration may enhance any physiological consequences.
• Bioblast editor: Gnaiger E
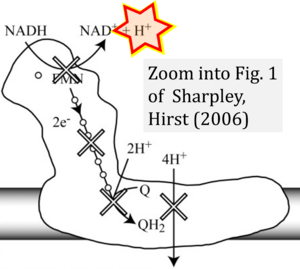
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels:
Enzyme: Complex I