Snyder 2009 Antioxid Redox Signal: Difference between revisions
From Bioblast
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Publication | {{Publication | ||
|title=Snyder CM, Chandel NS (2009) Mitochondrial regulation of cell survival and death during low-oxygen conditions. Antioxid Redox Signal 11:2673-83. doi | |title=Snyder CM, Chandel NS (2009) Mitochondrial regulation of cell survival and death during low-oxygen conditions. Antioxid Redox Signal 11:2673-83. https://doi.org/10.1089/ars.2009.2730 | ||
|info=[https://pubmed.ncbi.nlm.nih.gov/19580395/ PMID: 19580395 Open Access] | |info=[https://pubmed.ncbi.nlm.nih.gov/19580395/ PMID: 19580395 Open Access] | ||
|authors=Snyder CM, Chandel NS | |authors=Snyder CM, Chandel NS | ||
Latest revision as of 23:47, 23 April 2023
| Snyder CM, Chandel NS (2009) Mitochondrial regulation of cell survival and death during low-oxygen conditions. Antioxid Redox Signal 11:2673-83. https://doi.org/10.1089/ars.2009.2730 |
Snyder CM, Chandel NS (2009) Antioxid Redox Signal
Abstract: Mitochondria can initiate cell death or activate genes that promote cell survival in response to low oxygen. The BCL-2 family of proteins regulate cell death in response to anoxia (0-0.5% O2). By contrast, under hypoxia (0.5-3% O2), mitochondrial oxidative stress activates hypoxia-inducible factors (HIFs) to promote cell survival. In this review, we discuss how mitochondria, BCL-2 proteins, and HIFs are crucial for cellular responses to low oxygen.
• Bioblast editor: Gnaiger E
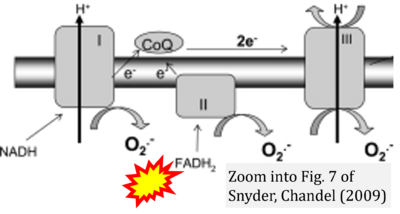
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels:
Stress:Hypoxia
Enzyme: Complex II;succinate dehydrogenase