Kaambre 2015 Abstract MiP2015
| Quantitative characterization of respiratory parameters of human colorectal and breast cancer clinical material. |
Link:
Shevchuk I, Koit A, Kaldma A, Chekulayev V, Klepinin A, Ounpuu L, Timohhina N, Tepp K, Kutner R, Heck K, Valvere V, Kaambre T (2015)
Event: MiP2015
A considerable part of previous studies about tumor bioenergetics were performed on several in vitro models with the conclusion that cancer cells present increased rates of glucose consumption and metabolize it to lactate even in the presence of O2 – a phenomenon called “Warburg effect”. In vitro studies cannot give the correct information about the functional activity and significance of OXPHOS versus glycolysis in malignancies and ignore host factors, which could exert significant effects. In our study we compare respiratory parameters of two very prevalent human tumors: breast cancer (HBC) and colorectal cancer (HCC).
Primary tumor samples were provided by the Oncology and Hematology Clinic at the North Estonia Medical Centre and were analysed immediately after surgery. In this work we investigated mitochondrial respiration of tumor and control tissues in situ using the skinned sample technique [1,2]. Rates of O2 consumption were assayed at 25 °C by an Oxygraph-2k high-resolution respirometer (Oroboros Instruments, Innsbruck Austria). The solubility of oxygen at 25 °C was taken as 240 nmol/ml. All respiration rates were normalized per mg dry weight of tissue.
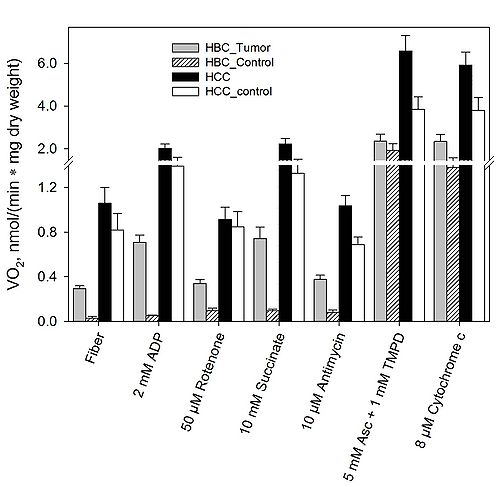
Multiple substrate-inhibitor titration protocol was used for the measurement of respiratory capacities of different respiratory chain (RC) segments (Fig. 1). To analyze these changes, the respiration rates for different RC complexes and ratios of respiration rates for different substrates were calculated. The HBC is not accompanied with suppression of complex I-dependent respiration as it is shown in colorectal cancer.
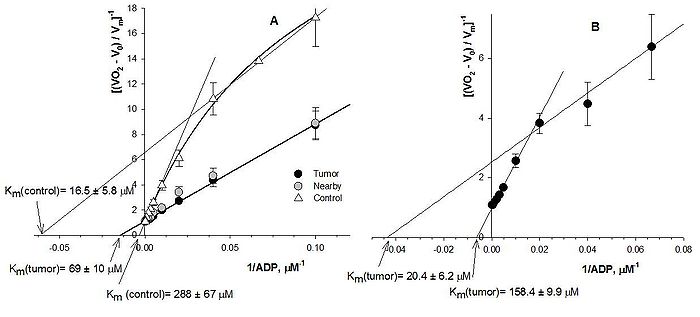
Apparent Michaelis-Menten constant (Km) and maximal rate of respiration (Vm) for ADP were calculated to characterize the affinity of mitochondria for exogenous ADP (permeability of mitochondrial outer membrane). Healthy colon tissue displayed low affinity for ADP (apparent Michaelis-Menten constant Km=256 ± 3 µM), whereas the affinity for ADP of tumor mitochondria (Km=93.6 ± 7.7 µM) and nearby tissue (junction area between cancer and normal mucosa) (Km=84.9 ± 9.9 µM) is significantly higher. Average Km value for HBC tissue samples was similar - 114.8±13.6 μM. Differences in Vmax correspond, to large extent, to the differences in number of mitochondria in these cell types. Measured rates of O2 consumption (normalized to Vm) were plotted vs. ADP concentration in medium as double reciprocal Lineweaver–Burk plots (Figure 2 A,B).
This data is showing that formation of colorectal cancer is associated with relative changes in the activities of individual respiratory chain complexes which may be the result of mitochondrial DNA mutations and/or variations in the assembly of respiratory chain supercomplexes.
Two subpopulations of mitochondria in HBC (Fig 2B) confirm the theory of two-compartment metabolism (“reversed Warburg”) proposed by several groups of cancer research [3,4]. During formation of HCC colon smooth muscle can participate in the carcinogenesis like energy reservoir and mitochondria lose the diffusion restrictions in the outer membrane. From all these results we can conclude that each type of cancer has its own special bioenergetic fingerprint.
• O2k-Network Lab: EE Tallinn Kaambre T, EE Tallinn Saks VA
Labels: MiParea: Respiration, Instruments;methods, mt-Membrane Pathology: Cancer
Organism: Human Tissue;cell: Endothelial;epithelial;mesothelial cell Preparation: Isolated mitochondria
Pathway: N HRR: Oxygraph-2k Event: A2, Poster, P-flash MiP2015
Affiliations
1-Lab Bioenergetics, Nat Inst Chemical Physics Biophysics; 2-Oncology Hematology Clinic North Estonia Medical Centre; 3-Tallinn Univ Technology; 4-Tallinn Univ, Inst Mathematics Natural Sciences; Tallinn, Estonia. - tuuli.kaambre@kbfi.ee
Figures
Figure 1. Respiratory chain analysis of HBC, HCC and healthy control breast and colon tissues.
Figure 2. Analysis of the dependence of the respiration rates on the exogenously added ADP in double reciprocal plots: HCC and two populations of mitochondria in control tissue (A) and two different populations of mitochondria in HBC (B).
References and acknowledgements
- Saks VA, Veksler VI, Kuznetsov AV, Kay L, Sikk P, Tiivel T, Tranqui L, Olivares J, Winkler K, Wiedemann F, Kunz WS (1998) Permeabilized cell and skinned fiber techniques in studies of mitochondrial function in vivo. Mol Cell Biochem 184:81-100.
- Kaambre T, Chekulayev V, Shevchuk I, Karu-Varikmaa M, Timohhina N, Tepp K, Bogovskaja J, Kütner R, Valvere V, Saks V (2012) Metabolic control analysis of cellular respiration in situ in intraoperational samples of human breast cancer. J Bioenerg Biomembr 44:539-58.
- Martinez-Outschoorn UE, Whitaker-Menezes D, Valsecchi M, Martinez-Cantarin MP, Dulau-Florea A, Gong J, Howell A, Flomenberg N, Pestell RG, Wagner J, Arana-Yi C, Sharma M, Sotgia F, Lisanti MP (2013) Reverse Warburg effect in a patient with aggressive B-cell lymphoma: is lactic acidosis a paraneoplastic syndrome? Semin Oncol 40:403-18.
- Witkiewicz AK, Whitaker-Menezes D, Dasgupta A, Philp NJ, Lin Z, Gandara R, Sneddon S, Martinez-Outschoorn UE, Sotgia F, Lisanti MP (2012) Using the "reverse Warburg effect" to identify high-risk breast cancer patients: stromal MCT4 predicts poor clinical outcome in triple-negative breast cancers. Cell Cycle 11:1108-17.