From Bioblast
| News and Events | Working Groups | Short-Term Scientific Missions | Management Committee | Members |
COST Action CA15203 (2016-2021): MitoEAGLE
Evolution-Age-Gender-Lifestyle-Environment: mitochondrial fitness mapping
MitoEAGLE data repository in muscle tissues
WG2
MitoEAGLE Working Groups Workshop, Obergurgl 2017
- MiPschool Obergurgl 2017 July 28-29.
WG2 - Steps towards database, July 28-29
- In the MitoEAGLE Working Groups Workshop (Obergurgl, 2017), the participants from the WG2 we discussed about the next steps for performing the data repository in muscle tissues. They were presented to all the participants. Here, a summary is showed: Summary from Obergurgl, 2017.
MitoFit Science Camp 3A: Skeletal and cardiac muscle fibres - towards a data repository
- MitoFit Science Camp 2016 Kuehtai AT Sunday, July 10
Circular 2016-06-15
- The first MitFit Science Camp (MitoFit SC) may be considered as a series of linked workshops, quite different from a conference or MiPschool. We would like to prepare with you the MitoFit-workshop on ‘Skeletal and cardiac muscle fibres – towards a data repository’.
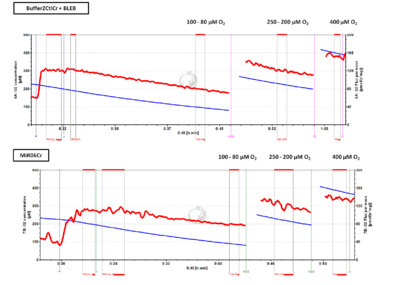
- Permeabilized muscle fibers (Pfi) are sensitive to low oxygen supply due to diffusion restrictions that limit mitochondrial respiration at the core of the fiber bundle. Therefore, hyperoxic conditions (oxygen range 500-250 μM) are required to counteract this limitation. Further studies have shown that the addition of a myosin II-specific inhibitor, blebbistatin (BLEB) in the respiration medium prevents fibre contraction, reduces the oxygen sensitivity and allows the study of ADP kinetics in Pfi at normoxic oxygen levels.
- In the Oroboros 'MitoFit lab', we recently evaluated ADP respiratory kinetics in skeletal muscle Pfi from mice under three different oxygen regimes at 37 °C: hypoxia (100-80 μM), normoxia (250-200 μM) and hyperoxia (400 μM) comparing two respiration media: MiR06+Cr (MiR06Cr) and Buffer Z with catalase (Ctl), creatine (Cr) and BLEB (BufferZCtlCr+BLEB). During her project as a visiting scientist, Nicole Bezuidenhout (O2k-Network lab ZA_Cape_Town_Ojuka_EO) observed a similar oxygen dependence in both media (Figure).
- Nicole will present her project at the MSC. Your input, results, experience, comments, suggestions are welcome to exchange views for understanding the oxygen dependence in permeabilized fibers from different samples. Adam Chicco has relevant data, we have data obtained by Hélène Lemieux during her postdoc project in Innsbruck, and would prefer a joint clarification and publication with your input, if possible.
- We have to consider many more topics for discussion, with the aim to take next steps towards presenting our data in a MitoEAGLE framework aimed at generating a common quantitative data base on mitochondrial respiratory function: normalization of respiration (wet weight, dry weight, mt-markers); SUIT protocols (a separate MitoFit-workshop at the beginning of the MSC, involving all participants); fibre preparation; comparison with isolated mitochondria and homogenate; statistical analysis (to be discussed during the last day of the MSC); ..
- We are looking forward to your contributions in the Skeletal and cardiac muscle fibres group at the MitoFit Science Camp 2016.
- With best regards,
- Erich, Carolina and the OROBOROS-team
- Erich Gnaiger, PhD, CEO
- Carolina Doerrier, PhD, CSO