Difference between revisions of "Magnesium Green"
| Line 1: | Line 1: | ||
{{MitoPedia | {{MitoPedia | ||
|abbr=MgG | |||
|description='''Magnesium green''' belongs to the [[extrinsic fluorophores]] applied for measurement of ATP production, based on different dissociation constants for ADP and ATP. | |description='''Magnesium green''' belongs to the [[extrinsic fluorophores]] applied for measurement of ATP production, based on different dissociation constants for ADP and ATP. | ||
}} | }} | ||
| Line 6: | Line 7: | ||
}} | }} | ||
{{MitoPedia topics}} | {{MitoPedia topics}} | ||
== Use of Magnesium Green 5N hexapotassium salt to measure ADP-ATP exchange rates == | |||
Contribution by [[Chinopoulos C|Christos Chinopoulos]], Semmelweis University, Budapest, Hungary | |||
'''Application of technique: ''' Online fluorescence measurement of ADP-ATP exchange | |||
<nowiki>[Mg<sup>2+</sup>]<sub>f</sub> </nowiki> determination from Magnesium Green (MgG) fluorescence in the extramitochondrial volume of isolated mitochondria and conversion to [[ADP]]-[[ATP]] exchange rate: Add mitochondria to 2 ml of an incubation medium of your choice. Including the adenylate kinase inhibitor Ap5A into the medium is essential; Mg<sup>2+</sup>, which is present in the assay medium, activates adenylate kinase. [[Ap5A]] is a potent inhibitor of [[adenylate kinase]]. MgG fluorescence can be recorded in a fluorometer, using 506 nm and 530 nm excitation and emission wavelengths, respectively. MgG exhibits an extremely high quantum yield <nowiki>(EM[MgG]=75,000 M-1*cm-1). At the end of each experiment, minimum fluorescence (Fmin) can be measured after addition of 4 mM EDTA, followed by the recording of maximum fluorescence (Fmax) elicited by addition of 20 mM MgCl<sub>2</sub>. Free Mg<sup>2+</sup> concentration, <nowiki>[Mg<sup>2+</sup>]<sub>f</sub></nowiki>, is calculated from the equation: <nowiki>[Mg<sup>2+</sup>]<sub>f</sub> </nowiki> = (''K''<sub>d</sub>(F-Fmin)/(Fmax-F))-0.055 mM, assuming a ''K''<sub>d</sub> of 0.9 mM for the MgG-Mg<sup>2+</sup> complex. The correction term -0.055 mM is empirical, and possibly reflects chelation of other ions by EDTA that have an affinity for MgG, and alter its fluorescence. This term is needed to obtain a reliable Mg<sup>2+</sup> estimate, as determined from calibration experiments using solutions with known, stepwise increasing, Mg<sup>2+</sup> concentrations. ADP-ATP exchange rate is estimated using the recently described method by our laboratory [1], exploiting the differential affinity of ADP and ATP to Mg<sup>2+</sup>. The rate of ATP appearing in the medium following addition of ADP to energized mitochondria (or vice versa in case of de-energized mitochondria), is calculated from the measured rate of change in free extramitochondrial <nowiki>[Mg<sup>2+</sup>]<sub>f</sub> </nowiki> using standard binding equations. The assay is designed such that the ANT is the sole mediator of changes in <nowiki>[Mg<sup>2+</sup>]<sub>f</sub> </nowiki> in the extramitochondrial volume, as a result of ADP-ATP exchange [1]. For the calculation of [ATP] or [ADP] from <nowiki>[Mg<sup>2+</sup>]<sub>f</sub> </nowiki>, the apparent ''K''<sub>d</sub> values should be estimated as described in [1].</nowiki> | |||
'''Validation of the technique''' | |||
'''Validation of the technique | |||
[[Image:Mg green checkpoints.jpg| 250px| right]] | [[Image:Mg green checkpoints.jpg| 250px| right]] | ||
| Line 25: | Line 25: | ||
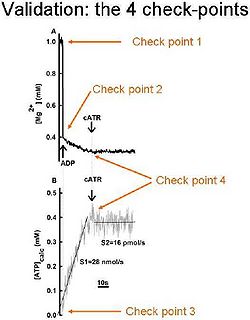
In order be sure that the calibrated MgG signal is converted correctly to ADP-ATP exchange rate, the following 4 check-points should be sought: | In order be sure that the calibrated MgG signal is converted correctly to ADP-ATP exchange rate, the following 4 check-points should be sought: | ||
# <nowiki>After calibration of the MgG signal to [Mg2+], | # <nowiki>After calibration of the MgG signal to [Mg2+], the initial [Mg2+]<sub>f</sub> value </nowiki>must be equal to that added in the cuvette by the experimenter; (i.e. did you add 1 mM MgCl2 in the cuvette? Then your calibrated MgG signal must start from 1 mM free Mg2+). | ||
# <nowiki>After addition of ADP (or ATP) to the medium, the [Mg2+]f value must drop to the expected level, after estimating the Kd of ADP (or ATP) for Mg2+; (i.e. if you estimated Kd of ADP for Mg2+ as 0.906 mM, the addition of 2 mM ADP to 1 mM Mg2+ leads to a free Mg2+ of 0.3939 mM).</nowiki> | # <nowiki>After addition of ADP (or ATP) to the medium, the [Mg2+]f value must drop to the expected level, after estimating the Kd of ADP (or ATP) for Mg2+; (i.e. if you estimated Kd of ADP for Mg2+ as 0.906 mM, the addition of 2 mM ADP to 1 mM Mg2+ leads to a free Mg2+ of 0.3939 mM).</nowiki> | ||
# <nowiki>After conversion on the calibrated [Mg2+]f to ADP, ATP level must start from “0”, if no ATP was already present in the medium, prior to adding ADP to mitochondria.</nowiki> | # <nowiki>After conversion on the calibrated [Mg2+]f to ADP, ATP level must start from “0”, if no ATP was already present in the medium, prior to adding ADP to mitochondria.</nowiki> | ||
| Line 33: | Line 33: | ||
<nowiki>[1] Chinopoulos C, Vajda S, Csanady L, Mandi M, Mathe K, | <nowiki>[1] Chinopoulos C, Vajda S, Csanady L, Mandi M, Mathe K, Adam-Vizi V (2009) A novel kinetic assay of mitochondrial ATP-ADP exchange rate mediated by the ANT. Biophys J 96: 2490-2504.</nowiki> | ||
Revision as of 10:15, 28 March 2012
Description
Magnesium green belongs to the extrinsic fluorophores applied for measurement of ATP production, based on different dissociation constants for ADP and ATP.
Abbreviation: MgG
MitoPedia methods:
Fluorometry
Use of Magnesium Green 5N hexapotassium salt to measure ADP-ATP exchange rates
Contribution by Christos Chinopoulos, Semmelweis University, Budapest, Hungary
Application of technique: Online fluorescence measurement of ADP-ATP exchange
[Mg<sup>2+</sup>]<sub>f</sub> determination from Magnesium Green (MgG) fluorescence in the extramitochondrial volume of isolated mitochondria and conversion to ADP-ATP exchange rate: Add mitochondria to 2 ml of an incubation medium of your choice. Including the adenylate kinase inhibitor Ap5A into the medium is essential; Mg2+, which is present in the assay medium, activates adenylate kinase. Ap5A is a potent inhibitor of adenylate kinase. MgG fluorescence can be recorded in a fluorometer, using 506 nm and 530 nm excitation and emission wavelengths, respectively. MgG exhibits an extremely high quantum yield (EM[MgG]=75,000 M-1*cm-1). At the end of each experiment, minimum fluorescence (Fmin) can be measured after addition of 4 mM EDTA, followed by the recording of maximum fluorescence (Fmax) elicited by addition of 20 mM MgCl<sub>2</sub>. Free Mg<sup>2+</sup> concentration, <nowiki>[Mg<sup>2+</sup>]<sub>f</sub>, is calculated from the equation: [Mg<sup>2+</sup>]<sub>f</sub> = (Kd(F-Fmin)/(Fmax-F))-0.055 mM, assuming a Kd of 0.9 mM for the MgG-Mg2+ complex. The correction term -0.055 mM is empirical, and possibly reflects chelation of other ions by EDTA that have an affinity for MgG, and alter its fluorescence. This term is needed to obtain a reliable Mg2+ estimate, as determined from calibration experiments using solutions with known, stepwise increasing, Mg2+ concentrations. ADP-ATP exchange rate is estimated using the recently described method by our laboratory [1], exploiting the differential affinity of ADP and ATP to Mg2+. The rate of ATP appearing in the medium following addition of ADP to energized mitochondria (or vice versa in case of de-energized mitochondria), is calculated from the measured rate of change in free extramitochondrial [Mg<sup>2+</sup>]<sub>f</sub> using standard binding equations. The assay is designed such that the ANT is the sole mediator of changes in [Mg<sup>2+</sup>]<sub>f</sub> in the extramitochondrial volume, as a result of ADP-ATP exchange [1]. For the calculation of [ATP] or [ADP] from [Mg<sup>2+</sup>]<sub>f</sub> , the apparent Kd values should be estimated as described in [1].</nowiki>
Validation of the technique
In order be sure that the calibrated MgG signal is converted correctly to ADP-ATP exchange rate, the following 4 check-points should be sought:
- After calibration of the MgG signal to [Mg2+], the initial [Mg2+]<sub>f</sub> value must be equal to that added in the cuvette by the experimenter; (i.e. did you add 1 mM MgCl2 in the cuvette? Then your calibrated MgG signal must start from 1 mM free Mg2+).
- After addition of ADP (or ATP) to the medium, the [Mg2+]f value must drop to the expected level, after estimating the Kd of ADP (or ATP) for Mg2+; (i.e. if you estimated Kd of ADP for Mg2+ as 0.906 mM, the addition of 2 mM ADP to 1 mM Mg2+ leads to a free Mg2+ of 0.3939 mM).
- After conversion on the calibrated [Mg2+]f to ADP, ATP level must start from “0”, if no ATP was already present in the medium, prior to adding ADP to mitochondria.
- The ADP-ATP exchange rate must be completely sensitive to carboxyatractyloside, a specific blocker of the ANT.
[1] Chinopoulos C, Vajda S, Csanady L, Mandi M, Mathe K, Adam-Vizi V (2009) A novel kinetic assay of mitochondrial ATP-ADP exchange rate mediated by the ANT. Biophys J 96: 2490-2504.