| Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. https://doi.org/10.26124/bec:2020-0002 |
Gnaiger Erich (2020) Bioenerg Commun
Abstract: ![]() https://doi.org/10.26124/bec:2020-0002
https://doi.org/10.26124/bec:2020-0002
Did you know that keeping your mitochondria fit is essential for quality of life, brain and muscle function, and resistance against preventable, immunological, and age-related degenerative diseases?
The capacity of cellular oxidative phosphorylation (OXPHOS) — a direct measure of mitochondrial function — is a result of evolution, age, gender, lifestyle, and environment (EAGLE). Increasingly, western lifestyle and aging contribute to mitochondrial dysfunction and the current epidemic of preventable diseases, including neurodegenerative and cardiovascular diseases, obesity, diabetes, and various types of cancer. The mitObesity epidemic leads to multimorbidity in aging and threatens to overwhelm the capacity of healthcare systems.
Training in mitochondrial physiology and bioenergetics, therefore, has high relevance to society. The ‘Blue Book’ on Mitochondrial Pathways and Respiratory Control presents a fundamental introduction to OXPHOS analysis for students and researchers in life sciences ― from evolutionary biology to medical and environmental applications. It combines concepts of bioenergetics and biochemical pathways related to mitochondrial core energy metabolism, provides the basis for substrate-uncoupler-inhibitor titration (SUIT) protocols, and updates the terminology consistent with the MitoEAGLE white paper on Mitochondrial Physiology.
It is now our responsibility to transfer the enthusiasm for innovation, reproducibility, and quality in science, and to translate mitochondrial research into visionary healthcare solutions.
• Keywords: Q-junction, Respiratory states, Flux control ratios, Additivity, Body mass excess
• Bioblast editor: Gnaiger E
• O2k-Network Lab: AT Innsbruck Gnaiger E, AT Innsbruck Oroboros
A guide through the chapters
- Real-time OXPHOS analysis. — Richard Altmann’s bioblasts are the systematic unit of bioenergetics and chemiosmotic coupling studied in living cells and mitochondrial preparations. A rigorous understanding of mitochondrial respiratory control relies on a clear concept of metabolic states and rates, accurate measurement and normalization of oxygen flux, and analysis of mitochondrial pathways.
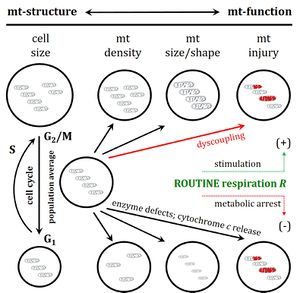
- Respiratory states and rates: coupling control. — A concept-driven terminology frames our perception of the meaning of respiratory states and rates, from ROUTINE respiration of living cells to the capacity of oxidative phosphorylation (OXPHOS) determined in mitochondrial preparations, electron transfer (ET) capacity, LEAK respiration, and the distinction of uncoupled, noncoupled, or dyscoupled respiration.
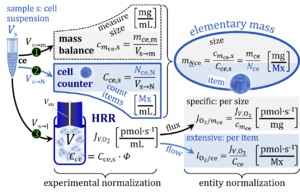
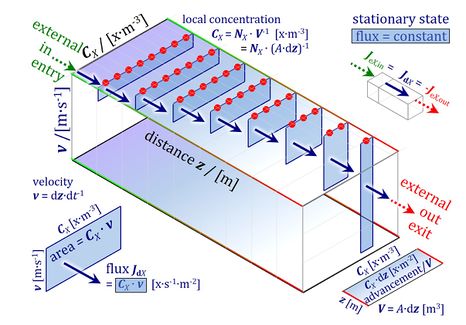
- Normalization of rate: flow, flux, and flux ratios. — ‘The challenges of measuring respiratory rate are matched by those of normalization’ (Gnaiger et al 2000). The effect of metabolic control variables on flow or flux can be expressed by normalization for rate in a reference state, and is evaluated relative to a background state. The concept of flux control efficiency is based on principles of thermodynamics and is guided by statistical considerations, to remove the bias of the classical respiratory control ratio.
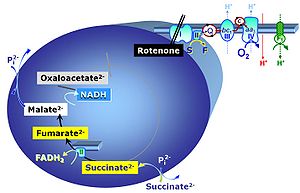
- NADH-linked pathways through Complex CI: respiratory pathway control with pyruvate, glutamate, malate. — Substrate combinations feeding electrons into the ET system through NADH have been considered to reflect physiological respiratory states in mitochondrial preparations. These protocols ignored the importance of cataplerotic metabolite depletion in the tricarboxylic acid (TCA) cycle.
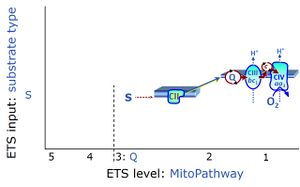
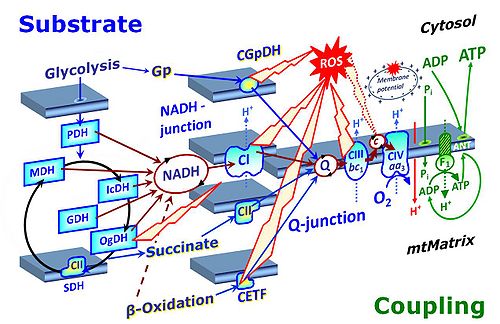
- S-pathway through Complex CII, F-pathway through CETF, Gp-pathway through CGpDH. — Succinate as the substrate of CII is at a level comparable to NADH as the substrate for CI. Too many textbooks and publications propagate the error of comparing NADH in the N-pathway with FADH2 in the S-pathway ― together with fumarate, FADH2 is a product but not a substrate of CII.
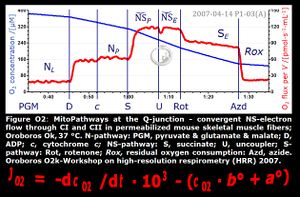
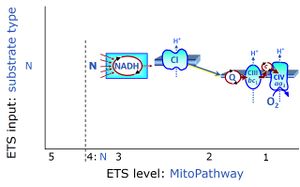
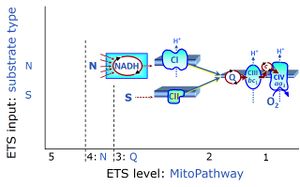
- NS-pathway through Complexes CI & CII: convergent electron transfer at the Q-junction. — The term ‘electron transport chain’ is a misnomer in bioenergetics, conceiling the convergent pathway architecture of the electron transfer system (ETS). This has direct implications on the design of substrate-uncoupler-inhibitor titration (SUIT) protocols, for reconstitution of TCA cycle function, and sequential separation of branches of mitochondrial pathways for OXPHOS analysis.
- Additivity of convergent electron transfer. — OXPHOS capacity depends on the degree of additivity of pathways converging at the Q-junction. Paradoxically, current concepts on interaction do not agree whether to categorize incompletely additive effects as synergistic or antagonistic. A new mathematical definition of additivity bridges the gap between these apparently incompatible models of interaction.
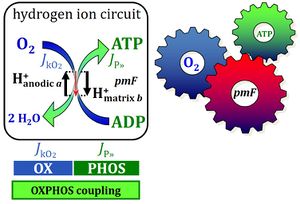
- Protonmotive pressure and respiratory control. — Why is thermodynamics scary? The driving force of chemical reactions is confusingly called an energy (Gibbs energy), whereas it is actually an isomorphic force, linked to the electric and chemical terms of the protonmotive force pmF. The gas law represents chemical force and gas pressure. Flux-force relations are non-linear. Why should we consider Fick’s linear law of diffusion and protonmotive pressure in the control of flux?
Preface
- Mitochondrial physiology is part of our lives. Mitochondrial fitness — the capacity of oxidative phosphorylation (OXPHOS) — is essential for the quality of your life, for brain and muscle function, for resistance against preventable and age-related degenerative diseases. Evolutionary background, age, gender (sex), lifestyle, and environmental factors (EAGLE) determine mitochondrial fitness, which is OXPHOS capacity and multiple mitochondrial functions. Comprehensive OXPHOS analysis is vital for understanding your cells, vital for our health care systems, and vitally deserves reliability and reproducibility of analytical and diagnostic studies.
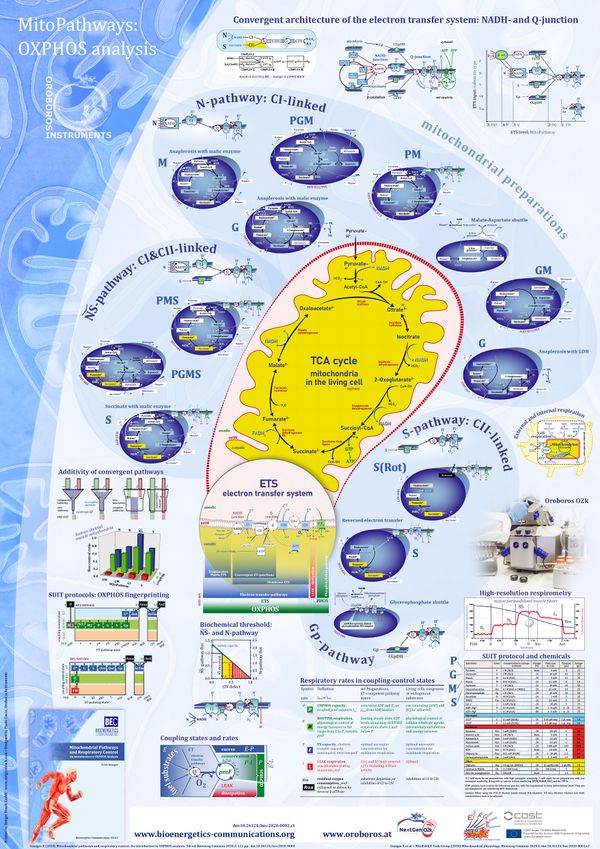
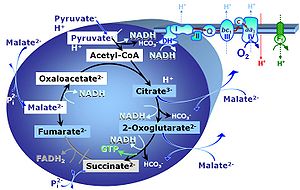
- The Blue Book on Mitochondrial Pathways and Respiratory Control presents a fundamental introduction to OXPHOS analysis. It combines concepts of bioenergetics and biochemical pathways related to mitochondrial (mt) core energy metabolism and provides the basis for the substrate-uncoupler-inhibitor titration (SUIT) protocols in high-resolution respirometry, which have been established since publication of the first edition of MitoPathways in 2007 (Figure 1).
- Application of SUIT protocols for real-time OXPHOS analysis is a component of metabolic phenotyping (Figure 2). OXPHOS analysis extends conventional bioenergetics to the level of mitochondrial physiology for functional diagnosis in health and disease. The Oroboros O2k for HRR has the high signal stability and unrestricted flexibility of titrations suited for application of elementary and complex SUIT protocols.
- Since 2007, research in mitochondrial physiology sparked a revolution of bioenergetics by experimental design that appreciates the convergent architecture of the electron transfer system (ETS) with multiple branches of mitochondrial pathways converging at the Q-junction, leading to a novel concept of additivity introduced in the new Chapter 7 of the Blue Book. These advancements are documented by >1 000 reports listed under 'NS-pathway control state' in MitoPedia. To study respiratory control at the Q-junction, SUIT protocols are applied with physiological substrate cocktails, particularly NADH-linked substrates (N) in combination with succinate (NS), fatty acids (FNS), and glycerophosphate (FNSGp), which have been introduced for the first time in the 1st edition of MitoPathways (2007).
- Since then, ‘MitoPedia’ was initiated and the COST Action MitoEAGLE flies. 666 coauthors joined forces to present a harmonized nomenclature on Mitochondrial Physiology (Bioenerg Commun 2020.1), with an emphasis on conceptual consistency for establishing a quality-controlled database on mitochondrial respiratory physiology. The 5th edition of MitoPathways gained from this collaboration. Many terms and symbols are simplified or presented in a more explicit form compared to the 2014 edition. Terms and iconic symbols develop meaning in context. Contextual meaning is best communicated by stories told in entertaining lectures, or by equations even if they turn off the most motivated student. Motivation is never enough. We need passion, persistence, resilience to transpose equations, terms and stories into the domain of personal experience, gaining perspective from perception to conception. The best scientific experience is the experiment driven by a hypothetical story written in clear words and forged into meaningful equations. This may provide a guideline to the critical discussion of the ergodynamic concept of the protonmotive force and chemiosmotic pressure, inspired by the Grey Book of Peter Mitchell and added as the new Chapter 8 of the Blue Book.
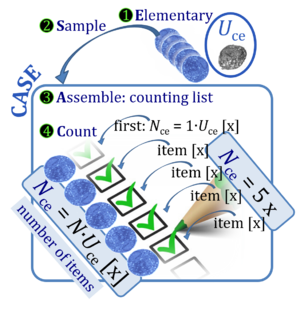
- Mitochondria are the structural and functional elementary units of cell respiration. MitoPathways is an element of the Oroboros Ecosystem driven by high-resolution respirometry and shaping mitochondrial physiology. A mosaic evolves by combining the elements into a picture of modern mitochondrial respiratory physiology.
- I thank all collaborators of the NextGen-O2k project and the authors and coauthors of various publications emerging from international cooperations, particularly the Horizon 2020 funded COST Action CA15203 MitoEAGLE. Without the team at Oroboros Instruments, including our partners in electromechanical engineering (O2k; WGT-Elektronik, Kolsass, Austria) and DatLab software development the experimental advances on MitoPathways would not have been possible.
- Erich Gnaiger
- Innsbruck, 2007 - 2020
Acknowledgements
- Specific thanks is extended to Oroboros team members Luiza Cardoso, Cristiane Cecatto, Carolina Doerrier, Sabine Schmitt, Timea Komlódi, Zulfiya Orynbayeva, and Lucie Zdrazilova for critical reading and helpful suggestions on various chapters, and to Univ.-Prof. Dr. Markus Haltmeier (Applied Mathematics, Univ Innsbruck, Austria) for stimulating discussions on additivity (Chapter 7).
Chapters: References and notes
- References Preface
- Gnaiger E (2014) Mitochondrial pathways and respiratory control. 4th ed. Oroboros MiPNet Publications, Innsbruck:80 pp. - »Bioblast link«
- Gnaiger E ed (2007) Mitochondrial pathways and respiratory control. 1st ed. Oroboros MiPNet Publications, Innsbruck:96 pp. - »Bioblast link«
- Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. https://doi.org/bec:2020-0001.v1
- References Preface
Chapter 1. Real-time OXPHOS analysis
References Chapter 1. OXPHOS
- Altmann R (1894) Die Elementarorganismen und ihre Beziehungen zu den Zellen. Zweite vermehrte Auflage. Verlag Von Veit & Comp, Leipzig:160 pp, 34 Tafeln. - »Bioblast link«
- Dawson KD, Baker DJ, Greenhaff PL, Gibala MJ (2005) An accute decrease in TCA cycle intermediates does not affect aerobic energy delivery in contracting rat skeletal muscle. - »Bioblast link«
- Garlid KD, Semrad C, Zinchenko V (1993) Does redox slip contribute significantly to mitochondrial respiration? - »Bioblast link«
- Gibala MJ, MacLean DA, Graham TE, Saltin B (1998) Tricarboxylic acid cycle intermediate pool size and estimated cycle flux in human muscle during exercise. - »Bioblast link« - Concentrations of TCA cycle intermediates.
- Gnaiger E (1983) Heat dissipation and energetic efficiency in animal anoxibiosis. Economy contra power. - »Bioblast link«
- Gnaiger E (1993) Efficiency and power strategies under hypoxia. Is low efficiency at high glycolytic ATP production a paradox? - »Bioblast link« - The Gibbs force of phorphorylation of ADP to ATP is FATP = 52 to 66 kJ/mol ATP under intracellular conditions.
- Gnaiger E (1993) Nonequilibrium thermodynamics of energy transformations. - »Bioblast link«
- Gnaiger E (2003) Oxygen conformance of cellular respiration. A perspective of mitochondrial physiology. - »Bioblast link«
- Gnaiger E (2008) Polarographic oxygen sensors, the oxygraph and high-resolution respirometry to assess mitochondrial function. - »Bioblast link«
- Gnaiger Erich (2020) Canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed. https://doi.org/doi:10.26124/mitofit:200004. -
- Gnaiger Erich et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. https://doi.org/10.26124/bec:2020-0001.v1. https://doi.org/10.26124bec2020-0001.v1
- Gnaiger E, Kuznetsov AV (2002) Mitochondrial respiration at low levels of oxygen and cytochrome c. - »Bioblast link«
- Gnaiger E, Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Steurer W, Margreiter R (2000b) Mitochondria in the cold. - »Bioblast link« – MiR05 as the basis of MiR06.
- Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. - »Bioblast link«
- Gnaiger E, Lassnig B, Kuznetsov AV, Rieger G, Margreiter R (1998) Mitochondrial oxygen affinity, respiratory flux control, and excess capacity of cytochrome c oxidase. - »Bioblast link«
- Gueguen N, Lefaucheur L, Ecolan P, Fillaut M, Herpin P (2005) Ca2+-activated myosin-ATPases, creatine and adenylate kinases regulate mitochondrial function according to myofibre type in rabbit. - »Bioblast link«
- Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer-pathway. XLII. Reconstitution of the electron transfer-pathway. - »Bioblast link«
- Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Mark W, Steurer W, Saks V, Usson Y, Margreiter R, Gnaiger E (2004) Mitochondrial defects and heterogeneous cytochrome c release after cardiac cold ischemia and reperfusion. - »Bioblast link« – Cytochrome c test.
- Lane N (2005) Power, sex, suicide: Mitochondria and the meaning of life. - »Bioblast link«
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. https://doi.org/10.1038/s41598-017-02789-8 - »Bioblast link«
- Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. - »Bioblast link«
- Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Glynn Research Ltd, Bodmin:192 pp. - »Bioblast link« - The Grey Book 1. - "or, writing Δp for the P.M.F." (p. 35)
- Mitchell P (1968) Chemiosmotic coupling and energy transduction. Glynn Research Ltd, Bodmin:111 pp. - The Grey Book 2.
- Mitchell P, Moyle J (1967) Respiration-driven proton translocation in rat liver mitochondria. Biochem J 105:1147-62. - »Bioblast link«
- Mootha VK, Arai AE, Balaban RS (1997) Maximum oxidative phosphorylation capacity of the mammalian heart. - »Bioblast link« – [Pi] <10 mM and [ADP] <0.4 mM limit OXPHOS in isolated heart mitochondria.
- Nicholson JK, Holmes E, Kinross JM, Darzi AW, Takats Z, Lindon JC (2012) Metabolic phenotyping in clinical and surgical environments. - »Bioblast link«
- Owen OE, Kalhan SC, Hanson RW (2002) The key role of anaplerosis and cataplerosis for citric acid cycle function. - »Bioblast link«
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopisies of human muscle. - »Bioblast link« - >90 % saturation is reached only >5 mM ADP, yet previously few studies used such high [ADP] in permeabilized tissues and cells. - Oxygen limitation of respiration below air saturation.
- Puchowicz MA, Varnes ME, Cohen BH, Friedman NR, Kerr DS, Hoppel CL (2004) Oxidative phosphorylation analysis: assessing the integrated functional activity of human skeletal muscle mitochondria – case studies. - »Bioblast link« - Cytochrome c test.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. - »Bioblast link« - Cytochrome c test.
- Renner K , Amberger A, Konwalinka G, Gnaiger E (2003) Changes of mitochondrial respiration, mitochondrial content and cell size after induction of apoptosis in leukemia cells. - »Bioblast link« -
- Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. - »Bioblast link«
- Saks VA, Veksler VI, Kuznetsov AV, Kay L, Sikk P, Tiivel T, Tranqui L, Olivares J, Winkler K, Wiedemann F, Kunz WS (1998) Permeabilised cell and skinned fiber techniques in studies of mitochondrial function in vivo. - »Bioblast link« - The apparent Km for ADP increases up to 0.5 mM in some permeabilized muscle fibres.
- Territo PR, Mootha VK, French SA, Balaban RS (2000) Ca2+ activation of heart mitochondrial oxidative phosphorylation: role of the FO/F1-ATPase. - »Bioblast link«
Notes: OXPHOS
Chapter 2. Respiratory states and rates: coupling control
References Chapter 2. States and rates
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. - »Bioblast link«
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. - »Bioblast link«
- Chance B, Williams GR (1956) The respiratory chain and oxidative phosphorylation. - »Bioblast link«
- Estabrook R (1967) Mitochondrial respiratory control and the polarographic measurement of ADP:O ratios. - »Bioblast link«
- Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. - »Bioblast link«
- Gnaiger Erich et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. https://doi.org/10.26124/bec:2020-0001.v1. - »Bioblast link«
- Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. - »Bioblast link« - Oxygen kinetics is different in the LEAK state without adenylates (LN) and State 4 (LEAK state with ATP, LN).
- Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. - »Bioblast link«
- König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. - »Bioblast link« - 3½ has been suggested to indicate an intermediate mitochondrial energy state somewhere between States 3 and 4. Would, therefore, State 4 be considered as being somewhere between State 3 and 5?
- Krumschnabel G, Eigentler A, Fasching M, Gnaiger E (2014) Use of safranin for the assessment of mitochondrial membrane potential by high-resolution respirometry and fluorometry. - »Bioblast link«
- Singh Simon (1997) Fermat's last theorem. Fourth Estate, London 340 pp. - »Bioblast link«
Notes: Coupling states
-
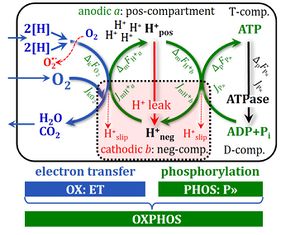
- A colour code is used with red and green in analogy to the states at a traffic light: at red, the motor is running in neutral gear (uncoupled) at minimum turnover without output (producing some heat) just to keep the engine running; at green, the motor is switched into gear and driven in a coupled state with full output. The blue colour is used to indicate a state of maximum input in neutral gear, or pressing fully the accelerator and the clutch simultaneously, which yields maximum turnover without output and produces a maximum of heat. The analogy for coupling in OXPHOS and in cars has its limitations but may help to memories the red/green colour code - you may think of it when your car is in a LEAK at the next red traffic light.
- H+ translocation through pumps is shown by dotted arrows across the mtIM.
-
Chapter 3. Normalization of rate: flow, flux, and flux ratios
References Chapter 3. Normalization
- Aguirre E, Rodríguez-Juárez F, Bellelli A, Gnaiger E, Cadenas S (2010) Kinetic model of the inhibition of respiration by endogenous nitric oxide in intact cells. - »Bioblast link« - Tables 3.1 and 3.2: HEK 293
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. - »Bioblast link«
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. - »Bioblast link«
- Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. - »Bioblast link«
- Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. - »Bioblast link«
- Hütter E, Renner K, Pfister G, Stöckl P, Jansen-Dürr P, Gnaiger E (2004) Senescence-associated changes in respiration and oxidative phosphorylation in primary human fibroblasts. - »Bioblast link« - Tables 3.1 and 3.2: fibrolasts; Figure 3.1.
- Hütter E, Unterluggauer H, Garedew A, Jansen-Dürr P, Gnaiger E (2006) High-resolution respirometry - a modern tool in aging research. - »Bioblast link«
- Renner K, Amberger A, Konwalinka G, Gnaiger E (2003) Changes of mitochondrial respiration, mitochondrial content and cell size after induction of apoptosis in leukemia cells. - »Bioblast link« - Tables 3.1 and 3.2: CEM
- Stadlmann S, Renner K, Pollheimer J, Moser PL, Zeimet AG, Offner FA, Gnaiger E (2006) Preserved coupling of oxidative phosphorylation but decreased mitochondrial respiratory capacity in IL-1ß treated human peritoneal mesothelial cells. - »Bioblast link«
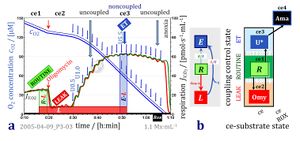
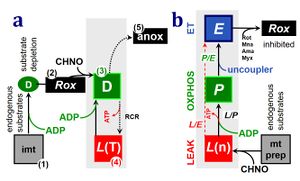
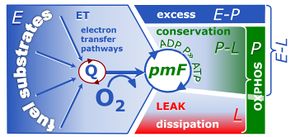
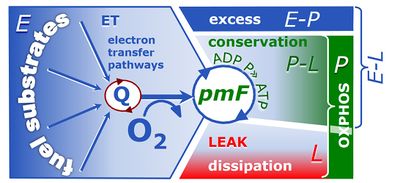
4-compartmental OXPHOS model. (1) ET capacity E of the noncoupled electron transfer system ETS. OXPHOS capacity P is partitioned into (2) the dissipative LEAK component L, and (3) ADP-stimulated P-L net OXPHOS capacity. (4) If P-L is kinetically limited by a low capacity of the phosphorylation system to utilize the protonmotive force pmF, then the apparent E-P excess capacity is available to drive coupled processes other than phosphorylation P» (ADP to ATP) without competing with P».
- Bioblast links: Coupling control - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
1. Mitochondrial and cellular respiratory rates in coupling-control states
| Respiratory rate | Defining relations | Icon | |
|---|---|---|---|
| OXPHOS capacity | P = P´-Rox | mt-preparations | |
| ROUTINE respiration | R = R´-Rox | living cells | |
| ET capacity | E = E´-Rox | » Level flow | |
| » Noncoupled respiration - Uncoupler | |||
| LEAK respiration | L = L´-Rox | » Static head | |
| » LEAK state with ATP | |||
| » LEAK state with oligomycin | |||
| » LEAK state without adenylates | |||
| Residual oxygen consumption Rox | L = L´-Rox |
2. Flux control ratios related to coupling in mt-preparations and living cells
| FCR | Definition | Icon | |
|---|---|---|---|
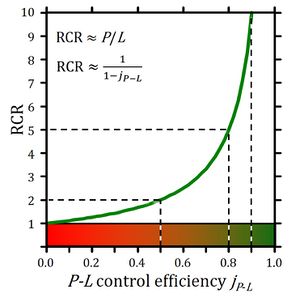
| L/P coupling-control ratio | L/P | » Respiratory acceptor control ratio, RCR = P/L | |
| L/R coupling-control ratio | L/R | ||
| L/E coupling-control ratio | L/E | » Uncoupling-control ratio, UCR = E/L (ambiguous) | |
| P/E control ratio | P/E | ||
| R/E control ratio | R/E | » Uncoupling-control ratio, UCR = E/L | |
| net P/E control ratio | (P-L)/E | ||
| net R/E control ratio | (R-L)/E |
3. Net, excess, and reserve capacities of respiration
| Respiratory net rate | Definition | Icon |
|---|---|---|
| P-L net OXPHOS capacity | P-L | |
| R-L net ROUTINE capacity | R-L | |
| E-L net ET capacity | E-L | |
| E-P excess capacity | E-P | |
| E-R reserve capacity | E-R |
4. Flux control efficiencies related to coupling-control ratios
| Coupling-control efficiency | Definition | Icon | Canonical term | ||
|---|---|---|---|---|---|
| P-L control efficiency | jP-L | = (P-L)/P | = 1-L/P | P-L OXPHOS-flux control efficiency | |
| R-L control efficiency | jR-L | = (R-L)/R | = 1-L/R | R-L ROUTINE-flux control efficiency | |
| E-L coupling efficiency | jE-L | = (E-L)/E | = 1-L/E | E-L ET-coupling efficiency » Biochemical coupling efficiency | |
| E-P control efficiency | jE-P | = (E-P)/E | = 1-P/E | E-P ET-excess flux control efficiency | |
| E-R control efficiency | jE-R | = (E-R)/E | = 1-R/E | E-R ET-reserve flux control efficiency |
5. General
- » Basal respiration
- » Cell ergometry
- » Dyscoupled respiration
- » Dyscoupling
- » Electron leak
- » Electron-transfer-pathway state
- » Hyphenation
- » Oxidative phosphorylation
- » Oxygen flow
- » Oxygen flux
- » Permeabilized cells
- » Phosphorylation system
- » Proton leak
- » Proton slip
- » Respiratory state
- » Uncoupling
Chapter 4. NADH-linked pathways through Complex I: respiratory pathway control with pruvate, glutamate, malate
References Chapter 4. N-pathways
- Brandt U (2006) Energy converting NADH:quinone oxidoreductase (Complex I). Annu Rev Biochem 75:69-92.
- Brewer GJ, Jones TT, Wallimann T, Schlattner U (2004) Higher respiratory rates and improved creatine stimulation in brain mitochondria isolated with antioxidants. - »Bioblast link«
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. - »Bioblast link« - Substrate depletion in isolated mitochondria is achieved in State 2: ADP is added to induce a transient stimulation of oxygen flux based on oxidation of endogenous substrates.
- Digerness SB, Reddy WJ (1976) The malate-aspartate shuttle in heart mitochondria. J Mol Cell Cardiol. 8:779-85.
- Duchen MR (2004) Roles of mitochondria in health and disease. Diabetes 53, Suppl 1:S96-102. - Mitochondrial glutamate dehydrogenase is particularly active in astrocytes, preventing glutamate induced neurotoxicity.
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. - »Bioblast link«
- Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. - »Bioblast link« - Equilibrium ratio of malate to fumarate is 4.1.
- Hildyard JCW, Halestrap AP (2003) Identification of the mitochondrial pyruvate carrier in Saccharomyces cerevidiae. Biochem J 374:607-11.
- Johnson G, Roussel D, Dumas JF, Douay O, Malthiery Y, Simard G, Ritz P (2006) Influence of intensity of food restriction on skeletal muscle mitochondrial energy metabolism in rats. Am J Physiol Endocrinol Metab 291:E460-7. - Uncoupling stimulates coupled OXPHOS respiration, PMP, by 14 %.
- Kemp RB, Hoare S, Schmalfeldt M, Bridge CM, Evans PM, Gnaiger E (1994) A thermochemical study of the production of lactate by glutaminolysis and glycolysis in mouse macrophage hybridoma cells. - »Bioblast link« - Glutamate derived from hydrolyzation of glutamine is a very important aerobic substrate in cultured cells.
- Lemasters JJ (1984) The ATP-to-oxygen stoichiometries of oxidative phosphorylation by rat liver mitochondria. - »Bioblast link« - Malonate added to inhibit the succinate-fumarate reaction exerts only a minor effect on liver mitochondrial respiration.
- Maechler P, Carobbio S, Rubi B (2006) In beta-cells, mitochondria integrate and generate metabolic signals controlling insulin secretion. Int J Biochem Cell Biol 38:696-709.
- Messer JI, Jackman MR, Willis WT (2004) Pyruvate and citric acid cycle carbon requirements in isolated skeletal muscle mitochondria. - »Bioblast link« - With malate alone and saturating [ADP] isolated rat skeletal muscle mitochondria respire at only 1.3 % of OXPHOS capacity with pyruvate+malate. Pyruvate alone yields only 2.1 % of OXPHOS capacity (P) with PM.
- Nicholls DG, Ferguson SJ (2002) Bioenergetics 3. - »Bioblast link« - Carriers.
- Ouhabi R, Boue-Grabot M, Mazat J-P (1994) ATP synthesis in permeabilized cells: Assessment of the ATP/O ratios in situ. - »Bioblast link« - In fibroblasts, GMP supports a higher respiratory flux than PMP.
- O’Donnell JM, Kudej RK, LaNoue KF, Vatner SF, Lewandowski ED (2004) Limited transfer of cytosolic NADH into mitochondria at high cardiac workload. Am J Physiol Heart Circ Physiol 286:H2237-42.
- Puchowicz MA, Varnes ME, Cohen BH, Friedman NR, Kerr DS, Hoppel CL (2004) Oxidative phosphorylation analysis: assessing the integrated functional activity of human skeletal muscle mitochondria – case studies. - »Bioblast link« - OXPHOS with glutamate alone is 50 % to 85 % of respiration with glutamate&malate. Accumulation of fumarate inhibits succinate dehydrogenase and glutamate dehydrogenase (Caughey et al 1957; Dervartanian, Veeger 1964). - OXPHOS with glutamate&malate is identical or 10 % higher than with pyruvate&malate.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. - »Bioblast link« - Uncoupling stimulates coupled OXPHOS respiration, PMP, by 15 % in human skeletal muscle. OXPHOS with glutamate alone is 50 % to 85 % of respiration with glutamate&malate. - OXPHOS with glutamate&malate is identical or 10 % higher than with pyruvate&malate.
- Schöpf B, Weissensteiner H, Schäfer G, Fazzini F, Charoentong P, Naschberger A, Rupp B, Fendt L, Bukur V, Giese I, Sorn P, Sant’Anna-Silva AC, Iglesias-Gonzalez J, Sahin U, Kronenberg F, Gnaiger E, Klocker H (2020) OXPHOS remodeling in high-grade prostate cancer involves mtDNA mutations and increased succinate oxidation. - »Bioblast link«
- Swenson ER (2018) Does aerobic respiration produce carbon dioxide or hydrogen ion and bicarbonate? - »Bioblast link«
- Thomas et al (2004) - OXPHOS in human skeletal muscle for PMP is 25 % higher than for GMP.
- Winkler-Stuck K, Kirches E, Mawrin C, Dietzmann K, Lins H, Wallesch CW, Kunz WS, Wiedemann FR (2005) Re-evaluation of the dysfunction of mitochondrial respiratory chain in skeletal muscle of patients with Parkinson's disease. - »Bioblast link« - OXPHOS in human skeletal muscle for PMP is 16% higher than for GMP.
Notes: N-pathways
- N-pathway control state
- Malic enzyme
- The metabolic maps in this and the following chapters have been modified and extended in comparison to previous editions. Added substrates are printed in blue in contrast to intermediates printed in black. CI-linked substrates and intermediates are shown with white background, whereas added succinate and consecutively formed fumarate are distinguished with a yellow background (FADH2 and the corresponding arrows are emphasized by yellow shades). Intermediates with grey background are considered to be present at low concentrations due to metabolite depletion, whereas products with blue background are considered to accumulate in the matrix space or in equilibrium with the large volume of incubation medium or to increase in equilibrium with the supplied substrate.
- Schwerzmann et al (1989) Proc Natl Acad Sci U S A 86:1583-7. - “Of the substrates used here, pyruvate/malate activates the chain at complex I, glutamate/malate and succinate at complexes II and III, ..” - This consideration of glutamate&malate requires correction.
- Ponsot et al (2005) J Cell Physiol 203:479-86. - (a) Respiration (State 3) in permeabilized fibres with malate alone gave 25-50 % of the flux with pyruvate+malate. This needs to be discussed in terms of endogenous mitochondrial substrates, which interfere to an unknown degree with the kinetics of respiration after addition of exogenous substrates, or the activity of malic enzyme. (b) Maximal respiration rates in muscle should be evaluated at saturating or high Pi, since at a Pi concentration of 3 mM OXPHOS respiration may be phosphate limited.
- Hulbert et al (2006) J Comp Physiol B 176:93-105. Addition of ‘sparking malate concentrations’. This term can probably be derived from the misconception that tricarboxylic acid cycle intermediates are conserved during respiration of isolated mitochondria. 380 µM malate in conjunction with 2.4 mM pyruvate were used, which makes a comparison difficult between different tissues and different species: the low substrate concentrations may limit PMP flux at various degrees in the different sources of mitochondria, and GMP or PGMP may support higher fluxes than PMP at tissue- and species-specific degrees.
Chapter 5. S-pathway through Complex II, F-pathway through electron-transferring flavoprotein, Gp-pathway through glycerophosphate dehydrogenase
References Chapter 5. S-, F-, Gp-pathways
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11. - With succinate alone OXPHOS is 30-40% lower than with succinate+rotenone in human skeletal muscle mitochondria.
- Cecchini G (2003) Function and structure of Complex II of the respiratory chain. Annu Rev Biochem 72:77-109.
- Ernster L, Nordenbrand K (1967) Skeletal muscle mitochondria. In: Estabrook RW, Pullman ME (eds) Meth Enzymol:86-94. – With succinate alone OXPHOS is 30-40% lower than with succinate+rotenone in rat skeletal muscle mitochondria.
- Jackman MR, Willis WT (1996) Characteristics of mitochondria isolated from type I and type IIb skeletal muscle. Am J Physiol Cell Physiol 270:C673-8. - Glycerophosphate oxidation is 10-fold higher in rabbit gracilis mitochondria compared to soleus.
- Lehninger AL (1970) Biochemistry. The molecular basis of cell structure and function Worth:833 pp. - Oxaloacetate is a more potent competitive inhibitor of succinate dehydrogenase than malonate even at small concentration (p 352).
- Muller FL, Liu Y, Abdul-Ghani MA, Lustgarten MS, Bhattacharya A, Jang YC, Van Remmen H (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. - »Bioblast link« - Addition of malate inhibits superoxide production with succinate, probably due to the oxaloacetate inhibition of CII.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. - »Bioblast link« – Glycerophosphate oxidation is relatively slow.
- Rauchova H, Drahota Z, Rauch P, Fato R, Lenaz G (2003) Coenzyme Q releases the inhibitory effect of free fatty acids on mitochondrial glycerophosphate dehydrogenase. Acta Biochim Polonica 50:405-13. - Glycerophosphate is an important substrate for respiration in brown adipose tissue mitochondria.
- Sun F, Huo X, Zhai Y, Wang A, Xu J, Su D, Bartlam M, Rao Z (2005) Crystal structure of mitochondrial respiratory membrane protein Complex II. Cell 121:1043–57.
- MitoPedia
Notes: S-, F-, Gp-pathways
- Succinate pathway
- Ponsot et al (2005) J Cell Physiol 203:479-86. - ‘.. the mitochondrial form of GPDH, which produces FADH2 within the mitochondrial matrix and provides electrons to Compoex II of the phosphorylation chain’. – The mitochondrial glycerophosphate dehydrogenase complex (CGpDH), located on the outer side of the inner mitochondrial membrane, does not provide electrons to CII, but feeds electrons into the Q-cycle entirely independent of CII. FADH2 is not produced within the mitochondrial matrix. Electron transfer takes place from the mitochondrial inner membrane flavoprotein-linked glycerophosphate dehydrogenase complex to CoQ.
Chapter 6. NS-pathway through Complexes CI & CII: convergent electron transfer at the Q-junction and additive effect of substrate combinations
References Chapter 6. Q-junction
- Aragonés J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, Dirkx R, Zacchigna S, Lemieux H, Jeoung NH, Lambrechts D, Bishop T, Lafuste P, Diez-Juan A, K Harten S, Van Noten P, De Bock K, Willam C, Tjwa M, Grosfeld A, Navet R, Moons L, Vandendriessche T, Deroose C, Wijeyekoon B, Nuyts J, Jordan B, Silasi-Mansat R, Lupu F, Dewerchin M, Pugh C, Salmon P, Mortelmans L, Gallez B, Gorus F, Buyse J, Sluse F, Harris RA, Gnaiger E, Hespel P, Van Hecke P, Schuit F, Van Veldhoven P, Ratcliffe P, Baes M, Maxwell P, Carmeliet P (2008) Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. - »Bioblast link« - OXPHOS analysis for phenotyping.
- Bianchi C, Genova ML, Parenti Castelli G, Lenaz G (2004) The mitochondrial respiratory chain is partially organized in a supercomplex assembly: kinetic evidence using flux control analysis. - »Bioblast link«
- Boushel R, Gnaiger E, Calbet JA, Gonzalez-Alonso J, Wright-Paradis C, Sondergaard H, Ara I, Helge JW, Saltin B (2011) Muscle mitochondrial capacity exceeds maximal oxygen delivery in humans. - »Bioblast link«
- Boushel R, Gnaiger E, Schjerling P, Skovbro M, Kraunsoe R, Flemming D (2007) Patients with Type 2 Diabetes have normal mitochondrial function in skeletal muscle. - »Bioblast link«
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11. - NS-substrate combination.
- Chance B (1965) Reaction of oxygen with the respiratory chain in cells and tissues. J Gen Physiol 49:163-88. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Costa LE, Boveris A, Koch OR, Taquini AC (1988) Liver and heart mitochondria in rats submitted to chronic hypobaric hypoxia. Am J Physiol Cell Physiol 255:C123-C9.
- Digerness SB, Reddy WJ (1976) The malate-aspartate shuttle in heart mitochondria. J Mol Cell Cardiol. 8:779-85.
- Eberhart K, Rainer J, Bindreither D, Ritter I, Gnaiger E, Kofler R, Oefner PJ, Renner K (2011) Glucocorticoid-induced alterations in mitochondrial membrane properties and respiration in childhood acute lymphoblastic leukemia. - »Bioblast link«
- Estabrook R (1967) Mitochondrial respiratory control and the polarographic measurement of ADP:O ratios. - »Bioblast link«
- Garait B, Couturier K, Servais S, Letexier D, Perrin D, Batandier C, Rouanet J-L, Sibille B, Rey B, Leverve X, Favier R (2005) Fat intake reverses the beneficial effects of low caloric intake on skeletal muscle mitochondrial H2O2 production. Free Radic Biol Med 39:1249–61. - GMP/GMSP substrate control ratio in skeletal muscle of rats fed on various diets ranges from 0.7 to 0.8.
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. - »Bioblast link«
- Gnaiger E, Boushel R, Søndergaard H, Munch-Andersen T, Damsgaard R, Hagen C, Díez-Sánchez C, Ara I, Wright-Paradis C, Schrauwen P, Hesselink M, Calbet JAL, Christiansen M, Helge JW, Saltin B (2015) Mitochondrial coupling and capacity of oxidative phosphorylation in skeletal muscle of Inuit and caucasians in the arctic winter. - »Bioblast link«
- Gnaiger E, Wright-Paradis C, Sondergaard H, Lundby C, Calbet JA, Saltin B, Helge J, Boushel R (2005) High-resolution respirometry in small biopsies of human muscle: correlations with body mass index and age. - »Bioblast link«
- González-Flecha B, Cutrin JC, Boveris A (1993) Time course and mechanism of oxidative stress and tissue damage in rat liver subjected to in vivo ischemia-reperfusion. J Clin Invest 91:456-64. - Respiration was measured in states GSP and GMP.
- Gutman M, Coles CJ, Singer TP, Casida JE (1971) On the functional organization of the respiratory chain at the dehydrogenase-coenzyme Q junction. Biochemistry 10:2036-43.
- Hansford RG, Hogue BA, Mildaziene V (1997) Dependence of H2O2 formation by rat heart mitochondria on substrate availability and donor age. J Bioenerg Biomembr 29:89–95.
- Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer-pathway. XLII. Reconstitution of the electron transfer-pathway. - »Bioblast link«
- König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. - »Bioblast link«
- Krebs HA (1935) CXCVII. Metabolism of amino-acids. III. Deamination of amino-acids. Biochem J 29:1620-44.
- Kunz WS, Kudin A, Vielhaber S, Elger CE, Attardi G, Villani G (2000) Flux control of cytochrome c oxidase in human skeletal muscle. - »Bioblast link«
- Kuznetsov AV, Clark JF, Winkler K, Kunz WS (1996) Increase of flux control of cytochrome c oxidase in copper-deficient mottled brindled mice. - »Bioblast link«
- Kuznetsov AV, Strobl D, Ruttmann E, Königsrainer A, Margreiter R, Gnaiger E (2002) Evaluation of mitochondrial respiratory function in small biopsies of liver. - »Bioblast link« - S(Rot) alone supports a higher flux than GM in liver mitocondria.
- Kuznetsov AV, Winkler K, Kirches E, Lins H, Feistner H, Kunz WS (1997) Application of inhibitor titrations for the detection of oxidative phosphorylation defects in saponin-skinned muscle fibers of patients with mitochondrial diseases. - »Bioblast link« - OXPHOS with glutamate&malate is identical or 10 % higher than with pyruvate&malate.
- LaNoue KF, Bryla J, Williamson JR (1972) Feedback interactions in the control of citric acid cycle activity in rat heart mitochondria. - »Bioblast link«
- LaNoue KF, Schoolwerth AC (1979) Metabolite transport in mitochondria. Annu Rev Biochem 48:871-922.
- Lehninger AL (1970) Biochemistry. The molecular basis of cell structure and function Worth:833 pp. - Electron transport chain.
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. - »Bioblast link«
- Lemieux H, Semsroth S, Antretter H, Hoefer D, Gnaiger E (2011) Mitochondrial respiratory control and early defects of oxidative phosphorylation in the failing human heart. - »Bioblast link« - SUIT protocols.
- Lenaz G, Genova ML (2009) Structural and functional organization of the mitochondrial respiratory chain: A dynamic super-assembly. Int J Biochem Cell Biol 41:1750-72.
- Llesuy S, Evelson P, González-Flecha B, Peralta J, Carreras MC, Poderoso JJ, Boveris A (1994) Oxidative stress in muscle and liver of rats with septic syndrome. Free Radic Biol Med 16:445-51. - GMP/GSP substrate control ratios are 0.8 and 0.7 in liver mitochondria (male Wistar and female Sprague-Dawley, respectively).
- Mogensen M, Sahlin K (2005) Mitochondrial efficiency in rat skeletal muscle: influence of respiration rate, substrate and muscle type. Acta Physiol Scand 185:229-36. - The flux control ratio of palmitoylcarnitine&malate/PM (OXPHOS) is 0.6 for mitochondria isolated from rat extensor digitorum longus muscle (mainly type II fibre type), but is 0.95 in rat soleus muscle (type I fibre type).
- Muller FL, Liu Y, Abdul-Ghani MA, Lustgarten MS, Bhattacharya A, Jang YC, Van Remmen H (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. - »Bioblast link«
- Nicholls DG, Ferguson SJ (2002) Bioenergetics 3. - »Bioblast link« - Electron transport chain.
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopisies of human muscle. - »Bioblast link« - SUIT protocols.
- Pesta D, Hoppel F, Macek C, Messner H, Faulhaber M, Kobel C, Parson W, Burtscher M, Schocke M, Gnaiger E (2011) Similar qualitative and quantitative changes of mitochondrial respiration following strength and endurance training in normoxia and hypoxia in sedentary humans. - »Bioblast link« - SUIT protocols.
- Rasmussen HN, Rasmussen UF (1997) Small scale preparation of skeletal muscle mitochondria, criteria for integrity, and assays with reference to tissue function. Mol Cell Biochem 174:55-60.
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. - »Bioblast link« - OXPHOS with palmitoylcarnitine&malate is 0.60 of OXPHOS with GM.
- Rasmussen UF, Rasmussen HN, Krustrup P, Quistorff B, Saltin B, Bangsbo J (2001) Aerobic metabolism of human quadriceps muscle: in vivo data parallel measurements on isolated mitochondria. - »Bioblast link«
- Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. - »Bioblast link«
- Rumsey WL, Schlosser C, Nuutinen EM, Robiolio M, Wilson DF (1990) Cellular energetics and the oxygen dependence of respiration in cardiac myocytes isolated from adult rat. J Biol Chem 265:15392-9. - 2.5-fold increase of respiration in the LEAK state with GMS compared to GM.
- Schägger H, Pfeiffer K (2001) The ratio of oxidative phosphorylation complexes I-V in bovine heart mitochondria and the composition of respiratory chain supercomplexes. J Biol Chem 276:37861-7.
- Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS (2005) Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A 102:5618-23.
- Simon J, van Spanning RJM, Richardson DJ (2008) The organisation of proton motive and non-proton motive redox loops in prokaryotic respiratory systems. Biochim Biophys Acta 1777:1480–90.
- Stadlmann S, Rieger G, Amberger A, Kuznetsov AV, Margreiter R, Gnaiger E (2002) H2O2-mediated oxidative stress versus cold ischemia-reperfusion: mitochondrial respiratory defects in cultured human endothelial cells. - »Bioblast link«
- Steinlechner-Maran R, Eberl T, Kunc M, Margreiter R, Gnaiger E (1996) Oxygen dependence of respiration in coupled and uncoupled endothelial cells. - »Bioblast link«
- Steinlechner-Maran R, Eberl T, Kunc M, Schröcksnadel H, Margreiter R, Gnaiger E (1997) Respiratory defect as an early event in preservation/reoxygenation injury in endothelial cells. - »Bioblast link«
- Sugano T, Oshino N, Chance B (1974). Mitochondrial functions under hypoxic conditions. The steady states of cytochrome c reduction and energy metabolism. Biochim Biophys Acta 347:340-58. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Tonkonogi M, Walsh B, Tiivel T, Saks V, Sahlin K (1999) Mitochondrial funciton in human skeletal muscle is not impaired by high intensity exercise. Eur J Physiol 437:562-8.
- Torres NV, Mateo F, Sicilia J, Meléndez-Hevia E. (1988) Distribution of the flux control in convergent metabolic pathways: theory and application to experimental and simulated systems. Int J Biochem 20:161-5.
- Villani G, Attardi G (1997) In vivo control of respiration by cytochrome c oxidase in wild-type and mitochondrial DNA mutation-carrying human cells. Proc Natl Acad Sci U S A 94:1166-71.
- Villani G, Greco M, Papa S, Attardi G (1998) Low reserve capacity of cytochrome c oxidase capacity in vivo in the respiratory chain of a variety of human cell types. - »Bioblast link«
- Votion DM, Gnaiger E, Lemieux H, Mouithys-Mickalad A, Serteyn D (2012) Physical fitness and mitochondrial respiratory capacity in horse skeletal muscle. - »Bioblast link« - SUIT protocols.
- Wiedemann FR, Winkler K, Kuznetsov AV, Bartels C, Vielhaber S, Feistner H, Kunz WS (1998) Impairment of mitochondrial function in skeletal muscle of patients with amyotrophic lateral sclerosis. - »Bioblast link« - OXPHOS with glutamate&malate is identical or 10% higher than with pyruvate&malate.
- Wilson DF, Rumsey WL, Green TJ, Vanderkooi J (1988). The oxygen dependence of mitochondrial oxidative phosphorylation measured by a new optical method for measuring oxygen concentration. J Biol Chem 263:2712-8. - Glutamate&succinate as respiratory substrate combination, without comparison of flux with different substrates.
- Zoccarato F, Cavallini L, Bortolami S, Alexandre A (2007) Succinate modulation of H2O2 release at NADH:ubiquinone oxidoreductase (Complex I) in brain mitochondria. Biochem J 406:125–9. - CI&II substrate combination.
Notes: Q-junction
- NS-pathway control state
- Identical GMP/GSP or GMP/GMSP ratios of 0.7 are reported for isolated mitochondria (Rasmussen, Rasmussen 2000; Capel et al 2005) and permeabilized fibers (Kunz et al 2000). For a review see Gnaiger (2009).
Chapter 7. Additivity of convergent electron transfer
References Chapter 7. Additivity
- Aragonés J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, et al. (2008) Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. - »Bioblast link«
- Avillac M, Hamed SB, Duhamel JR (2007) Multisensory integration in the ventral intraparietal area of the macaque monkey. J Neurosci 27:1922–32.
- Benard G, Bellance N, James D, Parrone P, Fernandez H, Letellier T, Rossignol R (2007) Mitochondrial bioenergetics and structural network organization. J Cell Sci:120:838-48.
- Berenbaum MC (1989) What is synergy? Pharmacol Rev 41:93-141.
- Boik JC, Newman RA, Boik RJ (2008) Quantifying synergism/antagonism using nonlinear mixed-effects modeling : A simulation study. Statistics in Medicine 27:1040-61.
- Boushel R, Gnaiger E, Schjerling P, Skovbro M, Kraunsøe R, Dela F (2007) Patients with Type 2 Diabetes have normal mitochondrial function in skeletal muscle. - »Bioblast link«
- Boyer PD (1993) The binding change mechanism for ATP synthase - Some probabilities and possibilities. Biochim Biophys Acta 1140:215-50.
- Brière JJ, Favier J, Gimenez-Roqueplo AP, Rustin P (2006) Tricarboxylic acid cycle dysfunction as a cause of human diseases and tumor formation. Am J Physiol Cell Physiol 291:C1114-20.
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11.
- Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. III. The steady state. - »Bioblast link«
- Chou TC, Talalay P (1977) A simple generalized equation for the analysis of multiple inhibitions of Michaelis-Menten kinetic systems. J Biol Chem 252:6438-42.
- Costa LE, Boveris A, Koch OR, Taquini AC (1988) Liver and heart mitochondria in rats submitted to chronic hypobaric hypoxia. Am J Physiol 255:C123-9.
- Elena SF, Lenski RE (1997) Test of synergistic interactions among deleterious mutations in bacteria. Nature 390:395–8.
- Fitzgerald J, Schoeberl B, Nielsen UB, Sorger PK (2006) Systems biology and combination therapy in the quest for clinical efficacy. Nat Chem Biol 2:458-66.
- Fritzen AJ, Grunnet N, Quistorff B (2007) Flux control analysis of mitochondrial oxidative phosphorylation in rat skeletal muscle: pyruvate and palmitoyl-carnitine as substrates give different control patterns. Eur J Appl Physiol 101:679–89.
- Garait B, Couturier K, Servais S, Letexier D, Perrin D, Batandier C, Rouanet J-L, Sibille B, Rey B, Leverve X, Favier R (2005) Fat intake reverses the beneficial effects of low caloric intake on skeletal muscle mitochondrial H2O2 production. Free Radic Biol Med 39:1249–61.
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. - »Bioblast link«
- Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. - »Bioblast link«
- Greco WR, Bravo G, Parsons JC (1995) The search for synergy: a critical review from a response surface perspective. Pharmacol Rev 47:331–85.
- Gutman M, Coles CJ, Singer TP, Casida JE (1971) On the functional organization of the respiratory chain at the dehydrogenase-coenzyme Q junction. - »Bioblast link«
- Gutman M, Silman N (1972) Mutual inhibition between NADH oxidase and succinoxidase activities in respiring submitochondrial particles. https://doi.org/10.1016/0014-5793(72)80574-x - »Bioblast link«
- Harris EJ, Manger JR (1968) Intramitochondrial substrate concentration as a factor controlling metabolism. The role of interanion competition. - »Bioblast link«
- Harris EJ, Manger JR (1969) Intersubstrate competitions and evidence for compartmentation in mitochondria. - »Bioblast link«
- Hartman JL, Garvik B, Hartwell L (2001) Cell biology - principles for the buffering of genetic variation. Science 291:1001–4.
- Haslam JM, Krebs HA (1963) Substrate competition in the respiration of animal tissues. The metabolic interactions of pyruvate and α-oxoglutarate in rat-liver homogenates. Biochem J 86:432-46.
- Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer system. XLII. Reconstitution of the electron transfer system. - »Bioblast link«
- Hegreness M, Shoresh N, Damian D, Hartl D, Kishony R (2008) Accelerated evolution of resistance in multidrug environments. Proc Natl Acad Sci USA 105:13977-81.
- Jaradat ZW, Zawistowski J (1996) Production and characterization of monoclonal antibodies against the O-5 antigen of Salmonella typhimurium lipopolysaccharide. Appl Environ Microbiol 62:1–5.
- Kalilani L, Atashili J (2006) Measuring additive interaction using odds ratios. Epidemiologic Perspectives Innovations 3:5. https://doi.org/10.1186/1742-5573-3-5
- Keilin D (1929) Cytochrome and respiratory enzymes. - »Bioblast link«
- King AJ, Palmer AR (1985) Integration of visual and auditory information in bimodal neurones in the guinea-pig superior colliculus. Exp Brain Res 60:492–500.
- König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. - »Bioblast link«
- Krebs HA (1940) The citric acid cycle. A reply to the criticisms of F. L. Breusch and of J. Thomas. Biochem J 34:460-3.
- Kunz WS, Kudin A, Vielhaber S, Elger CE, Attardi G, Villani G (2000) Flux control of cytochrome c oxidase in human skeletal muscle. - »Bioblast link«
- Lehár J, Zimmermann GR, Krueger AS, Molnar RA, Ledell JT, Heilbut AM, Short GF, Giusti LC, Nolan GP, Magid OA, Lee MS, Borisy AA, Stockwell BR, Keith CT (2007) Chemical combination effects predict connectivity in biological systems. Molecular Systems Biol 3:80.
- Lenaz G, Genova ML (2009) Structural and functional organization of the mitochondrial respiratory chain: A dynamic super-assembly. Int J Biochem Cell Biol 41:1750-72.
- Llesuy S, Evelson P, González-Flecha B, Peralta J, Carreras MC, Poderoso JJ, Boveris A (1994) Oxidative stress in muscle and liver of rats with septic syndrome. Free Radic Biol Med 16:445-51.
- Meredith MA, Stein BE (1983) Interactions among converging sensory inputs in the superior colliculus. Science 221:389–91.
- Michel JB, Yeh PJ, Chait R, Moellering RC Jr, Kishony R (2008) Drug interactions modulate the potential for evolution of resistance. Proc Natl Acad Sci USA 105:14918-23.
- Mitchell P (1979) Keilin’s respiratory chain concept and its chemiosmotic consequences. Science 206:1148-59.
- Natarajan M, Lin KM, Hsueh C, Sternweis PC, Ranganathan R (2006) A global analysis of cross-talk in a mammalian cellular signalling network. Nature Cell Biol 8:571-80.
- Patel Y, Gillet VJ, Howe T, Pastor J, Oyarzabal J, Willett P (2008) Assessment of additive/nonadditive effects in structure−activity relationships: Implications for iterative drug design. J Med Chem 51:7552–62.
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. - »Bioblast link«
- Rabøl R, Svendsen PF, Skovbro M, Boushel R, Haugaard SB, Schjerling P, Schrauwen P, Hesselink MK, Nilas L, Madsbad S, Dela F (2009) Reduced skeletal muscle mitochondrial respiration and improved glucose metabolism in nondiabetic obese women during a very low calorie dietary intervention leading to rapid weight loss. - »Bioblast link«
- Rasmussen HN, Rasmussen UF (1997) Small scale preparation of skeletal muscle mitochondria, criteria for integrity, and assays with reference to tissue function. - »Bioblast link«
- Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: A functional characterization. - »Bioblast link«
- Rasmussen UF, Rasmussen HN, Andersen AJ, Fogd Jorgensen P, Quistorff B (1996) Characterization of mitochondria from pig muscle: higher activity of exo-NADH oxidase in animals suffering from malignant hyperthermia. - »Bioblast link«
- Rasmussen UF, Rasmussen HN, Krustrup P, Quistorff B, Saltin B, Bangsbo J (2001) Aerobic metabolism of human quadriceps muscle: in vivo data parallel measurements on isolated mitochondria. - »Bioblast link«
- Rasmussen UF, Vielwerth SE, Rasmussen HN (2004) Skeletal muscle bioenergetics: a comparative study of mitochondria isolated from pigeon pectoralis, rat soleus, rat biceps brachii, pig biceps femoris and human quadriceps. - »Bioblast link«
- Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. - »Bioblast link«
- Schägger H, Pfeiffer K (2001) The ratio of oxidative phosphorylation complexes I-V in bovine heart mitochondria and the composition of respiratory chain supercomplexes. J Biol Chem 276:37861-7.
- Segrè D, DeLuna A, Church GM, Kishony R (2005) Modular epistasis in yeast metabolism. Nat Genet 37:77–83.
- Simon J, van Spanning RJM, Richardson DJ (2008) The organisation of proton motive and non-proton motive redox loops in prokaryotic respiratory systems. Biochim Biophys Acta 1777:1480–90.
- Stadlmann S, Renner K, Pollheimer J, Moser PL, Zeimet AG, Offner FA, Gnaiger E (2006) Preserved coupling of oxydative phosphorylation but decreased mitochondrial respiratory capacity in IL-1β treated human peritoneal mesothelial cells. - »Bioblast link«
- Steinlechner-Maran R, Eberl T, Kunc M, Schröcksnadel H, Margreiter R, Gnaiger E (1997) Respiratory defect as an early event in preservation/reoxygenation injury in endothelial cells. - »Bioblast link«
- Stock D, Leslie AGW, Walker JE (1999) Molecular architecture of the rotary motor in ATP synthase. Science 286:1700-05.
- Torella JP, Chait R, Kishony R (2010) Optimal drug synergy in antimicrobial treatments. https://doi.org/10.1371/journal.pcbi.1000796
- Torres NV, Mateo F, Sicilia J, Meléndez-Hevia E (1988) Distribution of the flux control in convergent metabolic pathways: theory and application to experimental and simulated systems. Int J Biochem 20:161-165.
- Tretiakov KV, Bishop KJM, Grzybowski BA (2009) Additivity of the excess energy dissipation rate in a dynamically self-assembled system. J Phys Chem B 113:7574–8.
- Villani G, Attardi G (1997) In vivo control of respiration by cytochrome c oxidase in wild-type and mitochondrial DNA mutation-carrying human cells. Proc Natl Acad Sci USA 94:1166-71.
- Wallace DC, Fan W, Procaccio V (2010) Mitochondrial energetics and therapeutics. Annu Rev Pathol 5:297-348.
- Yeh P, Tschumi AI, Kishony R (2006) Functional classification of drugs by properties of their pairwise interactions. Nat Genet 38:489-94.
- Yeh PJ, Hegreness MJ, Aiden AP, Kishony R (2009) Drug interactions and the evolution of antibiotic resistance. Nat Rev Microbiol 7:460–6.
Chapter 8. Protonmotive pressure and respiratory control
References Chapter 8. Protonmotive pressure
- Alberty RA, Daniels F (1980) Physical chemistry. SI version. 5th ed. - »Bioblast link«
- Bal W, Kurowska E, Maret W (2012) The final frontier of pH and the undiscovered country beyond. - »Bioblast link«
- Baum H (1967) Energetics of coupled events involving small compartments. - »Bioblast link«
- Beard DA (2005) A biophysical model of the mitochondrial respiratory system and oxidative phosphorylation. - »Bioblast link«
- Boltzmann L (1877) Über die Beziehung zwischen dem zweiten Hauptsatze der mechanischen Wärmetheorie und der Wahrscheinlichkeitsrechnung, respektive den Sätzen über das Wäremegleichgewicht. Sitzb d Kaiserlichen Akademie der Wissenschaften mathematisch-naturwissen Cl LXXVI, Abt II:373-435. - »Bioblast link«
- Boltzmann L (1896) Vorlesungen über Gastheorie I. Verlag Johann Ambrosius Barth, Leipzig:204 pp. - »Bioblast link«
- Bunnett JF, Jones RAY (1988) Names for hydrogen atoms, ions, and groups, and for reactions involving them. - »Bioblast link«
- Bureau International des Poids et Mesures (2019) The International System of Units (SI). 9th edition:117-216 ISBN 978-92-822-2272-0. - »Bioblast link«
- Canton M, Luvisetto S, Schmehl I, Azzone GF (1995) The nature of mitochondrial respiration and discrimination between membrane and pump properties. - »Bioblast link«
- Carnot Sadi (1824) Réflexions sur la puissance motrice du feu et sur les machines propres à développer cette puissance. Bachelier Paris. - »Bioblast link«
- Cohen ER, Cvitas T, Frey JG, Holmström B, Kuchitsu K, Marquardt R, Mills I, Pavese F, Quack M, Stohner J, Strauss HL, Takami M, Thor HL (2008) Quantities, Units and Symbols in Physical Chemistry. IUPAC Green Book 3rd Edition, 2nd Printing.- »Bioblast link«
- Coopersmith J (2010) Energy, the subtle concept. The discovery of Feynman’s blocks from Leibnitz to Einstein. - »Bioblast link«
- Divakaruni AS, Brand MD (2011) The regulation and physiology of mitochondrial proton leak. Physiology (Bethesda) 26:192-205.
- Dobbs BJT (1975) The foundations of Newton's alchemy or "The hunting of the Greene Lyon". Reissued as a paperback 1983. - »Bioblast link«
- Einstein A (1905) Über die von der molekularkinetischen Theorie der Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen. Ann Physik 4, XVII:549-60. - »Bioblast link« - The fundamental work of Fick 1855 Pogg Ann (Zurich) was not mentioned by Einstein (Zurich).
- Ferner RE, Aronson JK (2016) Cato Guldberg and Peter Waage, the history of the Law of Mass Action, and its relevance to clinical pharmacology. https://doi.org/10.1111/bcp.12721 - »Bioblast link«
- Fick Adolf (1855) Über Diffusion. Pogg Ann 94:59-86. - »Bioblast link«
- Garlid KD, Beavis AD, Ratkje SK (1989) On the nature of ion leaks in energy-transducing membranes. - »Bioblast link«
- Glasstone S (1948) Textbook of physical chemistry. 2nd ed, Macmillan and Co, London:1320 pp. - »Bioblast link« - The nomenclature which M Faraday used, and which is still employed, was devised for him by W. Whewell. Faraday assumed the flow of electricity to be associated with the movement of charged particles; these were called ions (Greek: wanderer).
- Gnaiger E (1989) Mitochondrial respiratory control: energetics, kinetics and efficiency. - »Bioblast link«
- Gnaiger E (1993) Efficiency and power strategies under hypoxia. Is low efficiency at high glycolytic ATP production a paradox? - »Bioblast link«
- Gnaiger E (1993) Nonequilibrium thermodynamics of energy transformations. - »Bioblast link«
- Gnaiger E (1994) Negative entropy for living systems: controversy between Nobel Laureates Schrödinger, Pauling and Perutz. - »Bioblast link«
- Gnaiger E (2020) Canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed. https://doi.org/10.26124/mitofit:200004.
- Gnaiger E, Forstner H, eds (1983) Polarographic oxygen sensors. Aquatic and physiological applications. Springer, Berlin, Heidelberg, New York:370 pp. - »Bioblast link«
- Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. - »Bioblast link«
- Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. https://doi.org/10.26124/bec:2020-0001.v1. - »Bioblast link«
- Grosholz ER (2007) Representation and productive ambiguity in mathematics and the sciences. Oxford Univ Press. - »Bioblast link«
- Headrick JM, Diken EG, Walters RS, Hammer NI, Christie RA, Cui J, Myshakin EM, Duncan MA, Johnson MA, Jordan KD (2005) Spectral signatures of hydrated proton vibrations in water clusters. Science 308:1765–69.
- Hitchman ML, Gnaiger E (1983) A thermodynamic consideration of permeability coefficients of membranes. - »Bioblast link«
- Kedem O, Katchalsky A (1958) Thermodynamic analysis of the permeability of biological membranes to non-electrolytes. - »Bioblast link« - "The misleading name ‘mean of the concentrations of the solute in the two compartments’ has been given to the term αX" (free activity).
- Kell DB (1979) On the functional proton current pathway of electron transport phosphorylation: An electrodic view. Biochim Biophys Acta 549:55-99.
- Klusch N, Murphy BJ, Mills DJ, Yildiz Ö, Kühlbrandt W (2017) Structural basis of proton translocation and force generation in mitochondrial ATP synthase. https://doi.org/10.7554/eLife.33274 - »Bioblast link«
- Komlódi T, Geibl FF, Sassani M, Ambrus A, Tretter L (2018) Membrane potential and delta pH dependency of reverse electron transport-associated hydrogen peroxide production in brain and heart mitochondria. - »Bioblast link«
- Laner V, Gnaiger E, eds (2014) Mitochondrial physiology – methods, concepts and biomedical perspectives. MiP2014. - »Bioblast link« - Mitchell's dream by Odra Noel [1]
- Maxwell JC (1867) On the dynamical theory of gases. - »Bioblast link«
- Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemiosmotic type of mechanism. - »Bioblast link« – Photo: [www.nobelprize.org/nobel_prizes/chemistry/laureates/1978/]
- Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. - »Bioblast link« – The Grey Book
- Mitchell P (1967) Proton current flow in mitochondrial systems. - »Bioblast link« - A discussion on the small number of protons in a mitochondrion, with emphasis on the membrane potential as the major component of the protonmotive force (p.m.f.).
- Mitchell P, Moyle J (1967) Respiration-driven proton translocation in rat liver mitochondria. - »Bioblast link«
- Mohr PJ, Phillips WD (2015) Dimensionless units in the SI. - »Bioblast link«
- Nernst W (1921) Studies in chemical thermodynamics. Nobel Lecture December 12, 1921. - »Bioblast link«
- Nicholls DG, Ferguson SJ (2013) Bioenergetics4. Academic Press. - »Bioblast link«
- Odra Noel, Gnaiger Erich (2014) MiPArt - Mitchell's dream - Mitchell's equation. - »Bioblast link«
- Onsager L (1931) Reciprocal relations in irreversible processes. I. - »Bioblast link«
- Patzek Tad W (2014) Fick’s diffusion experiments revisited — Part I. - »Bioblast link«
- Poburko Damon, Santo-Domingo Jaime, Demaurex Nicolas (2011) Dynamic regulation of the mitochondrial proton gradient during cytosolic calcium elevations. - »Bioblast link«
- Prebble J, Weber B (2003) Wandering in the gardens of the mind. Peter Mitchell and the making of Glynn. Oxford Univ Press.
- Prigogine I (1967) Introduction to thermodynamics of irreversible processes. Interscience, New York, 3rd ed. - »Bioblast link«
- Rich P (2003) Chemiosmotic coupling: The cost of living. - »Bioblast link«
- Rottenberg H (1984) Membrane potential and surface potential in mitochondria: uptake and binding of lipophilic cations. - »Bioblast link«
- Scaduto RC Jr, Grotyohann LW (1999) Measurement of mitochondrial membrane potential using fluorescent rhodamine derivatives. - »Bioblast link«
- Schrödinger E (1944) What is life? The physical aspect of the living cell. Cambridge Univ Press. - »Bioblast link«
- Schwerzmann K, Cruz-Orive LM, Eggman R, Sänger A, Weibel ER (1986) Molecular architecture of the inner membrane of mitochondria from rat liver: a combined biochemical and stereological study. - »Bioblast link«
- Swenson Erik R (2018) Does aerobic respiration produce carbon dioxide or hydrogen ion and bicarbonate? - »Bioblast link« - Facilitated diffusion: codiffusion with CO2 of bicarbonate and facilitated H+ transport by intracellular diffusion of buffer molecules.
- van't Hoff JH (1901) Osmotic pressure and chemical equilibrium. Nobel Lecture December 13, 1901. - »Bioblast link«
- Wang T (2010) Coulomb force as an entropic force. - »Bioblast link«
- White M (1997) Isaak Newton. The last sorcerer. Fourth Estate. - »Bioblast link«
A. Conversions
References Supplement A. Conversions
- Brooks GA, Hittelman KJ, Faulkner JA, Beyer RE (1971) Temperature, skeletal muscle mitochondrial functions, and oxygen debt. Am J Physiol 220:1053-9.
- Gnaiger E (1983) Symbols and units: Toward standardization. In: Polarographic Oxygen Sensors. Aquatic and Physiological Applications. Gnaiger E, Forstner H (eds), Springer, Berlin, Heidelberg, New York:352-8.
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. - »Bioblast link«
- Slater EC, Rosing J, Mol A (1973) The phosphorylation potential generated by respiring mitochondria. Biochim Biophys Acta 292:534-53.
B: SUIT
Substrates, uncouplers and inhibitors
Abbreviations
References
| Link | Reference | Year | View |
|---|---|---|---|
| Aguirre 2010 Biochim Biophys Acta | Aguirre E, Rodríguez-Juárez F, Bellelli A, Gnaiger E, Cadenas S (2010) Kinetic model of the inhibition of respiration by endogenous nitric oxide in intact cells. Biochim Biophys Acta 1797:557-65. https://doi.org/10.1016/j.bbabio.2010.01.033 | 2010 | PMID: 20144583 Open Access |
| Alberty 1980 Physical chemistry | Alberty RA, Daniels F (1980) Physical chemistry. SI version. 5th ed, John Wiley & Sons, New York:692 pp. | 1980 | |
| Altmann 1894 Verlag Von Veit & Comp | Altmann R (1894) Die Elementarorganismen und ihre Beziehungen zu den Zellen. Zweite vermehrte Auflage (The Elementary Organisms and Their Relationships to the Cells. Second Extended Edition). Verlag Von Veit & Comp, Leipzig. 160 pp, 34 Tafeln. | 1894 | books.google.com |
| Aragones 2008 Nat Genet | Aragonés J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, Dirkx R, Zacchigna S, Lemieux H, Jeoung NH, Lambrechts D, Bishop T, Lafuste P, Diez-Juan A, K Harten S, Van Noten P, De Bock K, Willam C, Tjwa M, Grosfeld A, Navet R, Moons L, Vandendriessche T, Deroose C, Wijeyekoon B, Nuyts J, Jordan B, Silasi-Mansat R, Lupu F, Dewerchin M, Pugh C, Salmon P, Mortelmans L, Gallez B, Gorus F, Buyse J, Sluse F, Harris RA, Gnaiger Erich, Hespel P, Van Hecke P, Schuit F, Van Veldhoven P, Ratcliffe P, Baes M, Maxwell P, Carmeliet P (2008) Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. Nat Genet 40:170-80. | 2008 | PMID: 18176562; PMID: 18176562 Open Access;
» |
| Avillac 2007 J Neurosci | Avillac M, Hamed SB, Duhamel JR (2007) Multisensory integration in the ventral intraparietal area of the macaque monkey. J Neurosci 27:1922–32. | 2007 | |
| Bal 2012 PLOS ONE | Bal W, Kurowska E, Maret W (2012) The final frontier of pH and the undiscovered country beyond. PLOS ONE 7(9):e45832. | 2012 | https://doi.org/10.1371/journal.pone.0045832 |
| Baum 1967 Nature | Baum H (1967) Energetics of coupled events involving small compartments. Nature 214:1326–7. | 1967 | https://www.nature.com/articles/2141326a0 |
| Beard 2005 PLOS Comput Biol | Beard DA (2005) A biophysical model of the mitochondrial respiratory system and oxidative phosphorylation. PLoS Comput Biol 1(4):e36. | 2005 | PMID: 16163394 Open Access |
| Benard 2007 J Cell Sci | Benard G, Bellance N, James D, Parrone P, Fernandez H, Letellier T, Rossignol R (2007) Mitochondrial bioenergetics and structural network organization. J Cell Sci 120:838-48. | 2007 | |
| Berenbaum 1989 Pharmacol Rev | Berenbaum MC (1989) What is synergy?. Pharmacol Rev 41:93-141. | 1989 | |
| Bianchi 2004 J Biol Chem | Bianchi C, Genova ML, Parenti Castelli G, Lenaz G (2004) The mitochondrial respiratory chain is partially organized in a supercomplex assembly: kinetic evidence using flux control analysis. J Biol Chem 279:36562-9. | 2004 | PMID: 15205457 Open Access |
| Boik 2008 Statistics in Medicine | Boik JC, Newman RA, Boik RJ (2008) Quantifying synergism/antagonism using nonlinear mixed-effects modeling : A simulation study. Statistics in Medicine 27:1040-61. | 2008 | |
| Boltzmann 1877 Sitzb d Kaiserlichen Akademie | Boltzmann L (1877) Über die Beziehung zwischen dem zweiten Hauptsatze der mechanischen Wärmetheorie und der Wahrscheinlichkeitsrechnung, respektive den Sätzen über das Wäremegleichgewicht. Sitzb d Kaiserlichen Akademie der Wissenschaften mathematisch-naturwissen Cl LXXVI, Abt II:373-435. | 1877 | |
| Boltzmann 1896 Verlag Johann Ambrosius Barth, Leipzig | Boltzmann Ludwig (1896) Vorlesungen über Gastheorie I. Verlag Johann Ambrosius Barth, Leipzig:204 pp. | 1896 | Open Access |
| Boushel 2011 Mitochondrion | Boushel RC, Gnaiger E, Calbet JA, Gonzalez-Alonso J, Wright-Paradis C, Sondergaard H, Ara I, Helge JW, Saltin B (2011) Muscle mitochondrial capacity exceeds maximal oxygen delivery in humans. Mitochondrion 11:303-7. | 2011 | PMID: 21147270 |
| Boushel 2007 Diabetologia | Boushel RC, Gnaiger E, Schjerling P, Skovbro M, Kraunsoee R, Dela F (2007) Patients with Type 2 diabetes have normal mitochondrial function in skeletal muscle. Diabetologia 50:790-6. | 2007 | PMID: 17334651 Open Access |
| Boyer 1993 Biochim Biophys Acta | Boyer PD (1993) The binding change mechanism for ATP synthase - Some probabilities and possibilities. Biochim Biophys Acta 1140:215-50. | 1993 | |
| Brandt 2006 Annu Rev Biochem | Brandt U (2006) Energy converting NADH:quinone oxidoreductase (Complex I). Annu Rev Biochem 75:69-92. | 2006 | |
| Brewer 2004 Mitochondrion | Brewer GJ, Jones TT, Wallimann T, Schlattner U (2004) Higher respiratory rates and improved creatine stimulation in brain mitochondria isolated with anti-oxidants. Mitochondrion 4:49-57. | 2004 | PMID: 16120374 |
| Briere 2006 Am J Physiol Cell Physiol | Brière JJ, Favier J, Gimenez-Roqueplo AP, Rustin P (2006) Tricarboxylic acid cycle dysfunction as a cause of human diseases and tumor formation. Am J Physiol Cell Physiol 291:C1114-20. | 2006 | |
| Brooks 1971 Am J Physiol | Brooks GA, Hittelman KJ, Faulkner JA, Beyer RE (1971) Temperature, skeletal muscle mitochondrial functions, and oxygen debt. Am J Physiol 220:1053-9. | 1971 | |
| Bunnett 1988 Pure Appl Chem | Bunnett JF, Jones RAY (1988) Names for hydrogen atoms, ions, and groups, and for reactions involving them. Pure Appl Chem 60:1115-16. | 1988 | Open Access |
| Bureau International des Poids et Mesures 2019 The International System of Units (SI) | Bureau International des Poids et Mesures (2019) The International System of Units (SI). 9th edition:117-216. ISBN 978-92-822-2272-0 | 2019 | »Open Access« |
| Canton 1995 Biochem J | Canton M, Luvisetto S, Schmehl I, Azzone GF (1995) The nature of mitochondrial respiration and discrimination between membrane and pump properties. Biochem J 310:477-81. | 1995 | PMID: 7654185 Open Access |
| Capel 2005 Mech Ageing Develop | Capel F, Rimbert V, Lioger D, Diot A, Rousset P, Patureau Mirand P, Boirie Y, Morio B, Mosoni L (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Develop 126:505-11. | 2005 | |
| Carnot 1824 Bachelier | Carnot Sadi (1824) Réflexions sur la puissance motrice du feu et sur les machines propres à développer cette puissance. Bachelier Paris. | 1824 | WorldCat |
| Cecchini 2003 Annu Rev Biochem | Cecchini G (2003) Function and structure of Complex II of the respiratory chain. Annu Rev Biochem 72:77-109. https://doi.org/10.1146/annurev.biochem.72.121801.161700 | 2003 | PMID: 14527321 |
| Chance 1952 Nature | Chance B (1952) Spectra and reaction kinetics of respiratory pigments of homogenized and intact cells. Nature 169:215-21. | 1952 | PMID: 14910730 |
| Chance 1965 J Gen Physiol | Chance B (1965) Reaction of oxygen with the respiratory chain in cells and tissues. https://doi.org/10.1085/jgp.49.1.163 | 1965 | J Gen Physiol 49:163-88. PMID: 4285727 Open Access |
| Chance 1955 J Biol Chem-III | Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation: III. The steady state. J Biol Chem 217:409-27. | 1955 | PMID: 13271404 Open Access |
| Chance 1955 J Biol Chem-I | Chance B, Williams GR (1955) Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. J Biol Chem 217:383-93. | 1955 | PMID: 13271402 Open Access |
| Chance 1956 Adv Enzymol Relat Subj Biochem | Chance B, Williams GR (1956) The respiratory chain and oxidative phosphorylation. Adv Enzymol Relat Subj Biochem 17:65-134. | 1956 | PMID: 13313307 |
| Chou 1977 J Biol Chem | Chou TC, Talalay P (1977) A simple generalized equation for the analysis of multiple inhibitions of Michaelis-Menten kinetic systems. J Biol Chem 252:6438-42. | 1977 | |
| Cohen 2008 IUPAC Green Book | Cohen ER, Cvitas T, Frey JG, Holmström B, Kuchitsu K, Marquardt R, Mills I, Pavese F, Quack M, Stohner J, Strauss HL, Takami M, Thor HL (2008) Quantities, Units and Symbols in Physical Chemistry. IUPAC Green Book 3rd Edition, 2nd Printing, IUPAC & RSC Publishing, Cambridge. | 2008 | Open Access |
| Coopersmith 2010 Oxford Univ Press | Coopersmith J (2010) Energy, the subtle concept. The discovery of Feynman’s blocks from Leibnitz to Einstein. Oxford Univ Press:400 pp. | 2010 | |
| Costa 1988 Am J Physiol | Costa LE, Boveris A, Koch OR, Taquini AC (1988) Liver and heart mitochondria in rats submitted to chronic hypobaric hypoxia. Am J Physiol 255:C123-9. | 1988 | |
| Crabtree 1929 Biochem J | Crabtree HG (1929) Observations on the carbohydrate metabolism of tumours. Biochem J 23:536–45. | 1929 | PMC1254097 |
| Dawson 2005 J Physiol | Dawson KD, Baker DJ, Greenhaff PL, Gibala MJ (2005) An accute decrease in TCA cycle intermediates does not affect aerobic energy delivery in contracting rat skeletal muscle. J Physiol 565:637-43. | 2005 | PMID: 15802296 |
| Digerness 1976 J Mol Cell Cardiol | Digerness SB, Reddy WJ (1976) The malate-aspartate shuttle in heart mitochondria. J Mol Cell Cardiol 8:779-85. | 1976 | |
| Divakaruni 2011 Physiology (Bethesda) | Divakaruni AS, Brand MD (2011) The regulation and physiology of mitochondrial proton leak. Physiology (Bethesda) 26:192-205. | 2011 | |
| Dobbs 1975 Cambridge Univ Press | Dobbs BJT (1975) The foundations of Newton's alchemy or "The hunting of the Greene Lyon". Reissued as a paperback 1983. Cambridge Univ Press Cambridge:300 pp. | 1975 | |
| Doerrier 2018 Methods Mol Biol | Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. https://doi.org/10.1007/978-1-4939-7831-1_3 | 2018 | PMID: 29850993 » |
| Duchen 2004 Diabetes | Duchen MR (2004) Roles of mitochondria in health and disease. Diabetes 53, Suppl 1:S96-102. | 2004 | |
| Eberhart 2011 Biochim Biophys Acta | Eberhart K, Rainer J, Bindreither D, Ritter I, Gnaiger E, Kofler R, Oefner PJ, Renner K (2011) Glucocorticoid-induced alterations in mitochondrial membrane properties and respiration in childhood acute lymphoblastic leukemia. Biochim Biophys Acta 1807:719-25. | 2011 | PMID: 21237131 Open Access |
| Einstein 1905 Ann Physik 549 | Einstein A (1905) Über die von der molekularkinetischen Theorie der Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen. Ann Physik 4, XVII:549-60. | 1905 | Open Access |
| Elena 1997 Nature | Elena SF, Lenski RE (1997) Test of synergistic interactions among deleterious mutations in bacteria. Nature 390:395–8. | 1997 | |
| Ernster 1967 Meth Enzymol | Ernster L, Nordenbrand K (1967) Skeletal muscle mitochondria. Meth Enzymol Estabrook RW, Pullman ME (eds):86-94. | 1967 | |
| Ernster 1981 J Cell Biol | Ernster L, Schatz G (1981) Mitochondria: a historical review. J Cell Biol 91:227s-55s. | 1981 | PMID: 7033239 Open Access |
| Estabrook 1967 Methods Enzymol | Estabrook RW (1967) Mitochondrial respiratory control and the polarographic measurement of ADP:O ratios. Methods Enzymol 10:41-7. | 1967 | ScienceDirect |
| Ferner 2016 Br J Clin Pharmacol | Ferner RE, Aronson JK (2016) Cato Guldberg and Peter Waage, the history of the Law of Mass Action, and its relevance to clinical pharmacology. Br J Clin Pharmacol 81:52-5. doi:10.1111/bcp.12721. | 2016 | Open Access |
| Fick 1855 Pogg Ann | Fick Adolf (1855) Über Diffusion. Pogg Ann 94:59-86. | 1855 | Open Access |
| Fitzgerald 2006 Nat Chem Biol | Fitzgerald J, Schoeberl B, Nielsen UB, Sorger PK (2006) Systems biology and combination therapy in the quest for clinical efficacy. Nat Chem Biol 2:458-66. | 2006 | |
| Fritzen 2007 Eur J Appl Physiol | Fritzen AJ, Grunnet N, Quistorff B (2007) Flux control analysis of mitochondrial oxidative phosphorylation in rat skeletal muscle: pyruvate and palmitoyl-carnitine as substrates give different control patterns. Eur J Appl Physiol 101:679–89. | 2007 | |
| Gallavotti 2014 Springer | Gallavotti G (2014) Nonequilibrium and irreversibility. Springer Heidelberg, New York, London. | 2014 | |
| Garait 2005 Free Radic Biol Med | Garait B, Couturier K, Servais S, Letexier D, Perrin D, Batandier C, Rouanet J-L, Sibille B, Rey B, Leverve X, Favier R (2005) Fat intake reverses the beneficial effects of low caloric intake on skeletal muscle mitochondrial H2O2 production. Free Radic Biol Med 39:1249–61. | 2005 | |
| Garlid 1989 Biochim Biophys Acta | Garlid KD, Beavis AD, Ratkje SK (1989) On the nature of ion leaks in energy-transducing membranes. Biochim Biophys Acta 976:109-20. | 1989 | PMID: 2675980 |
| Garlid 1993 BTK | Garlid KD, Semrad C, Zinchenko V (1993) Does redox slip contribute significantly to mitochondrial respiration? In: Schuster S, Rigoulet M, Ouhabi R, Mazat J-P (eds) Modern trends in biothermokinetics. Plenum Press, New York, London:287-93. | 1993 | |
| Gibala 1998 Am J Physiol Endocrinol Metab | Gibala MJ, MacLean DA, Graham TE, Saltin B (1998) Tricarboxylic acid cycle intermediate pool size and estimated cycle flux in human muscle during exercise. Am J Physiol Endocrinol Metab 275:E235-42. | 1998 | PMID: 9688624 Open Access |
| Glasstone 1948 Macmillan and Co, London | Glasstone Samuel (1948) Textbook of physical chemistry. 2nd ed, Macmillan and Co, London:1320 pp. | 1948 | |
| Gnaiger 1983 J Exp Zool | Gnaiger E (1983) Heat dissipation and energetic efficiency in animal anoxibiosis. Economy contra power. J Exp Zool 228:471-90. | 1983 | |
| Gnaiger 1983 POS symbols | Gnaiger E (1983) Symbols and units: toward standardization. In: Polarographic Oxygen Sensors. Aquatic and Physiological Applications. Gnaiger E, Forstner H (eds), Springer, Berlin, Heidelberg, New York:352-8. | 1983 | |
| Gnaiger 1989 Energy Transformations | Gnaiger E (1989) Mitochondrial respiratory control: energetics, kinetics and efficiency. In: Energy transformations in cells and organisms. Wieser W, Gnaiger E (eds), Thieme, Stuttgart:6-17. | 1989 | |
| Gnaiger 1993 Hypoxia | Gnaiger E (1993) Efficiency and power strategies under hypoxia. Is low efficiency at high glycolytic ATP production a paradox? In: Surviving hypoxia: Mechanisms of control and adaptation. Hochachka PW, Lutz PL, Sick T, Rosenthal M, Van den Thillart G (eds) CRC Press, Boca Raton, Ann Arbor, London, Tokyo:77-109. | 1993 | |
| Gnaiger 1993 Pure Appl Chem | Gnaiger E (1993) Nonequilibrium thermodynamics of energy transformations. Pure Appl Chem 65:1983-2002. http://dx.doi.org/10.1351/pac199365091983 | 1993 | PAC:199365091983 Open Access PAC |
| Gnaiger 2001 Respir Physiol | Gnaiger E (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. https://doi.org/10.1016/S0034-5687(01)00307-3 | 2001 | Respir Physiol 128:277-97. PMID: 11718759 |
| Gnaiger 2003 Adv Exp Med Biol | Gnaiger E (2003) Oxygen conformance of cellular respiration. A perspective of mitochondrial physiology. https://doi.org/10.1007/978-1-4419-8997-0_4 | 2003 | Adv Exp Med Biol 543:39-55. PMID: 14713113 |
| Gnaiger 2008 POS | Gnaiger E (2008) Polarographic oxygen sensors, the oxygraph and high-resolution respirometry to assess mitochondrial function. In: Mitochondrial dysfunction in drug-induced toxicity (Dykens JA, Will Y, eds) John Wiley & Sons, Inc, Hoboken, NJ:327-52. | 2008 | |
| Gnaiger 2009 Int J Biochem Cell Biol | Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. https://doi.org/10.1016/j.biocel.2009.03.013 | 2009 | PMID: 19467914 |
| Gnaiger 2017 MiP2017 | Gnaiger E (2017) Protonmotive force and chemiosmotic pressure: a generalization of non-ohmic flux control of the proton leak. MiP2017. | ||
| Gnaiger 2020 MitoFit x | Gnaiger E (2021) The elementary unit — canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed. https://doi.org/10.26124/mitofit:200004.v2 | 2021 | MitoFit Preprints 2020.04.v2. The elementary unit — canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed. |
| BEC 2020.1 doi10.26124bec2020-0001.v1 | Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. https://doi.org/10.26124/bec:2020-0001.v1 | 2020 | Bioenerg Commun 2020.1. |
| Gnaiger 2015 Scand J Med Sci Sports | Gnaiger E, Boushel R, Søndergaard H, Munch-Andersen T, Damsgaard R, Hagen C, Díez-Sánchez C, Ara I, Wright-Paradis C, Schrauwen P, Hesselink M, Calbet JAL, Christiansen M, Helge JW, Saltin B (2015) Mitochondrial coupling and capacity of oxidative phosphorylation in skeletal muscle of Inuit and caucasians in the arctic winter. https://doi.org/10.1111/sms.12612 | 2015 | Scand J Med Sci Sports 25 (Suppl 4):126–34. PMID: 26589126 Open Access » |
| Gnaiger 1983 Springer POS | Gnaiger E, Forstner H, eds (1983) Polarographic Oxygen Sensors. Aquatic and Physiological Applications. Springer, Berlin, Heidelberg, New York:370 pp. | 1983 | Springer link |
| Gnaiger 2002 Biochem Soc Trans | Gnaiger E, Kuznetsov AV (2002) Mitochondrial respiration at low levels of oxygen and cytochrome c. Biochem Soc Trans 30:242-8. | 2002 | PMID 12023860 |
| Gnaiger 2000 Life in the Cold | Gnaiger E, Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Steurer W, Margreiter R (2000) Mitochondria in the cold. In: Life in the Cold (Heldmaier G, Klingenspor M, eds) Springer, Berlin, Heidelberg:431-42. https://doi.org/10.1007/978-3-662-04162-8_45 | 2000 | |
| Gnaiger 1998 Biochim Biophys Acta | Gnaiger E, Lassnig B, Kuznetsov AV, Margreiter R (1998) Mitochondrial respiration in the low oxygen environment of the cell: Effect of ADP on oxygen kinetics. Biochim Biophys Acta 1365:249-54. https://doi.org/10.1016/S0005-2728(98)00076-0 | 1998 | PMID: 9693739 Open Access |
| Gnaiger 1998 J Exp Biol | Gnaiger E, Lassnig B, Kuznetsov AV, Rieger G, Margreiter R (1998) Mitochondrial oxygen affinity, respiratory flux control, and excess capacity of cytochrome c oxidase. https://doi.org/10.1242/jeb.201.8.1129 | 1998 | Open Access J Exp Biol 201:1129-39. PMID: 9510525 |
| Gnaiger 2000 Proc Natl Acad Sci U S A | Gnaiger E, Méndez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. Proc Natl Acad Sci U S A 97:11080-5. https://doi.org/10.1073/pnas.97.20.11080 | 2000 | PMID: 11005877 Open Access |
| Gnaiger 1995 J Bioenerg Biomembr | Gnaiger E, Steinlechner-Maran R, Méndez G, Eberl T, Margreiter R (1995) Control of mitochondrial and cellular respiration by oxygen. https://doi.org/10.1007/BF02111656 | 1995 | J Bioenerg Biomembr 27:583-96. PMID: 8746845 |
| Gnaiger 1994 BTK-207 | Gnaiger E, Wyss M (1994) Chemical forces in the cell: Calculation for the ATP system. In: What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds) Modern Trends in BioThermoKinetics 3. Innsbruck Univ Press:207-12. | 1994 | |
| Gnaiger 2005 Abstract MiP2005 | Gnaiger Erich, Wright-Paradis C, Sondergaard H, Lundby C, Calbet Jose AL, Saltin Bengt, Helge J, Boushel R (2005) High-resolution respirometry in small biopsies of human muscle: correlations with body mass index and age. MiP2005. | ||
| Gonzalez-Flecha 1993 J Clin Invest | González-Flecha B, Cutrin JC, Boveris A (1993) Time course and mechanism of oxidative stress and tissue damage in rat liver subjected to in vivo ischemia-reperfusion. J Clin Invest 91:456-64. | 1993 | |
| Greco 1995 Pharmacol Rev | Greco WR, Bravo G, Parsons JC (1995) The search for synergy: a critical review from a response surface perspective. Pharmacol Rev 47:331–85. | 1995 | |
| Grosholz 2007 Oxford Univ Press | Grosholz Emily R (2007) Representation and productive ambiguity in mathematics and the sciences. Oxford Univ Press 312 pp. | 2007 | |
| Gueguen 2005 J Physiol | Gueguen N, Lefaucheur L, Ecolan P, Fillaut M, Herpin P (2005) Ca2+-activated myosin-ATPases, creatine and adenylate kinases regulate mitochondrial function according to myofibre type in rabbit. J Physiol 564:723-35. | 2005 | PMID: 15731190 Open Access |
| Gutman 1971 Biochemistry | Gutman M, Coles CJ, Singer TP, Casida JE (1971) On the functional organization of the respiratory chain at the dehydrogenase-coenzyme Q junction. Biochemistry 10:2036-43. | 1971 | |
| Gutman 1972 FEBS Lett | Gutman M, Silman N (1972) Mutual inhibition between NADH oxidase and succinoxidase activities in respiring submitochondrial particles. FEBS Lett 26:207-10. doi: 10.1016/0014-5793(72)80574-x. | 1972 | PMID: 4404628 Open Access |
| Goebel 2019 Wiley-VCH | Göbel EO, Siegner U (2019) The new international system of units (SI). Quantum metrology and quantum standards. Wiley-VCH 249 pp. | 2019 | books.google |
| Hansford 1997 J Bioenerg Biomembr | Hansford RG, Hogue BA, Mildaziene V (1997) Dependence of H2O2 formation by rat heart mitochondria on substrate availability and donor age. J Bioenerg Biomembr 29:89–95. | 1997 | |
| Harman 1952 Exp Cell Res | Harman JW, Feigelson M (1952) Studies on mitochondria IV. The cytological localization of mitochondria in heart muscle. Exp Cell Res 3:58-64. | 1952 | doi:10.1016/0014-4827(52)90031-1 |
| Harris 1968 Biochem J | Harris EJ, Manger JR (1968) Intramitochondrial substrate concentration as a factor controlling metabolism. The role of interanion competition. Biochem J 109:239-46. | 1968 | Open Access |
| Harris 1969 Biochem J | Harris EJ, Manger JR (1969) Intersubstrate competitions and evidence for compartmentation in mitochondria. Biochem J 113:617-28. | 1969 | |
| Hartman 2001 Science | Hartman JL, Garvik B, Hartwell L (2001) Cell biology - principles for the buffering of genetic variation. Science 291:1001–4. | 2001 | |
| Haslam 1963 Biochem J | Haslam JM, Krebs HA (1963) Substrate competition in the respiration of animal tissues. The metabolic interactions of pyruvate and α-oxoglutarate in rat-liver homogenates. Biochem J 86:432-46. | 1963 | |
| Hatefi 1962 J Biol Chem-XLII | Hatefi Y, Haavik AG, Fowler LR, Griffiths DE (1962) Studies on the electron transfer system XLII. Reconstitution of the electron transfer system. J Biol Chem 237:2661-9. https://doi.org/10.1016/S0021-9258(19)73804-6 | 1962 | PMID:13905326 Open Access |
| Hatefi 1962 J Biol Chem-XLI | Hatefi Y, Haavik AG, Griffiths DE (1962) Studies on the electron transfer system. XLI. Reduced coenzyme Q (QH2)-cytochrome c reductase. J Biol Chem 237:1681-5. | 1962 | PMID: 13905328 Open Access; PDF |
| Headrick 2005 Science | Headrick JM, Diken EG, Walters RS, Hammer NI, Christie RA, Cui J, Myshakin EM, Duncan MA, Johnson MA, Jordan KD (2005) Spectral signatures of hydrated proton vibrations in water clusters. Science 308:1765–69. | 2005 | |
| Hegreness 2008 Proc Natl Acad Sci U S A | Hegreness M, Shoresh N, Damian D, Hartl D, Kishony R (2008) Accelerated evolution of resistance in multidrug environments. Proc Natl Acad Sci U S A 105:13977-81. | 2008 | |
| Hildyard 2003 Biochem J | Hildyard JC, Halestrap AP (2003) Identification of the mitochondrial pyruvate carrier in Saccharomyces cerevisiae. Biochem J 374: 607-611. | 2003 | |
| Hitchman 1983 POS Membrane | Hitchman ML, Gnaiger E (1983) A thermodynamic consideration of permeability coefficients of membranes. In: Polarographic Oxygen Sensors. Aquatic and Physiological Applications. Gnaiger E, Forstner H (eds), Springer, Berlin, Heidelberg, New York:31-6. | 1983 | |
| Hochachka 2002 Oxford Univ Press | Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford Univ Press, New York: 466 pp. | 2002 | |
| Hofstadter 1979 Harvester Press | Hofstadter DR (1979) Gödel, Escher, Bach: An eternal golden braid. A metaphorical fugue on minds and machines in the spirit of Lewis Carroll. Harvester Press:499 pp. | 1979 | https://en.wikipedia.org/wiki/G%C3%B6del_Escher_Bach |
| Hulbert 2006 J Comp Physiol B | Hulbert AJ, Turner N, Hinde J, Else P, Guderley H (2006) How might you compare mitochondria from different tissues and different species?. J Comp Physiol B 176:93-105. | 2006 | PMID: 16408229 |
| Huetter 2004 Biochem J | Hütter E, Renner K, Pfister G, Stöckl P, Jansen-Dürr P, Gnaiger E (2004) Senescence-associated changes in respiration and oxidative phosphorylation in primary human fibroblasts. https://doi.org/10.1042/BJ20040095 | 2004 | Biochem J 380:919-28. PMID: 15018610 - Open Access |
| Huetter 2006 Exp Gerontol | Hütter E, Unterluggauer H, Garedew A, Jansen-Dürr P, Gnaiger E (2006) High-resolution respirometry - a modern tool in aging research. Exp Gerontol 41:103-9. | 2006 | PMID: 16309877 |
| Jackman 1996 Am J Physiol Cell Physiol | Jackman MR, Willis WT (1996) Characteristics of mitochondria isolated from type I and type IIb skeletal muscle. Am J Physiol Cell Physiol 270:C673-8. | 1996 | |
| Jaradat 1996 Appl Environ Microbiol | Jaradat ZW, Zawistowski J (1996) Production and characterization of monoclonal antibodies against the O-5 antigen of Salmonella typhimurium lipopolysaccharide. Appl Environ Microbiol 62:1–5. | 1996 | |
| Johnson 2006 Am J Physiol Endocrinol Metab | Johnson G, Roussel D, Dumas JF, Douay O, Malthiery Y, Simard G, Ritz P (2006) Influence of intensity of food restriction on skeletal muscle mitochondrial energy metabolism in rats. Am J Physiol Endocrinol Metab 291:E460-7. | 2006 | |
| Jung 1944 Walter-Verlag | Jung CG (1944) Psychologie und Alchemie. Walter-Verlag Olten, 2. Auflage 1976:620 pp. ISBN 3-530-40712-7. | 1944 | |
| Kalilani 2006 Epidemiologic Perspectives Innovations | Kalilani L, Atashili J (2006) Measuring additive interaction using odds ratios. Epidemiologic Perspectives Innovations 3:5 doi:10.1186/1742-5573-3-5. | 2006 | |
| Kedem 1958 Biochim Biophys Acta | Kedem O, Katchalsky A (1958) Thermodynamic analysis of the permeability of biological membranes to non-electrolytes. Biochim Biophys Acta 27:229-46. | 1958 | PMID: 13522722 |
| Keilin 1925 Proc R Soc Lond B Biol Sci | Keilin D (1925) On cytochrome, a respiratory pigment, common to animals, yeast, and higher plants. Proc R Soc Lond B Biol Sci 98:312-39. | 1925 | |
| Keilin 1929 Proc R Soc London Ser B | Keilin D (1929) Cytochrome and respiratory enzymes. Proc R Soc London Ser B 104:206-52. | 1929 | http://www.jstor.org/pss/81339 |
| Kell 1979 Biochim Biophys Acta | Kell DB (1979) On the functional proton current pathway of electron transport phosphorylation: An electrodic view. Biochim Biophys Acta 549:55-99. | 1979 | |
| King 1985 Exp Brain Res | King AJ, Palmer AR (1985) Integration of visual and auditory information in bimodal neurones in the guinea-pig superior colliculus. Exp Brain Res 60:492–500. | 1985 | |
| Klingenberg 1960 Biochem Z | Klingenberg M, Schollmeyer P (1960) On the reversibility of oxidative phosphorylation. Adenosine triphosphate-dependent respiratory control and reduction of diphosphopyridine nucleotide in mitochondria. Biochem Z 333:335-51. | 1960 | PMID: 13756883 |
| Klusch 2017 Elife | Klusch N, Murphy BJ, Mills DJ, Yildiz Ö, Kühlbrandt W (2017) Structural basis of proton translocation and force generation in mitochondrial ATP synthase. Elife e33274. doi: 10.7554/eLife.33274. | 2017 | PMID: 29210357 Open Access |
| Komlodi 2018 J Bioenerg Biomembr | Komlódi T, Geibl FF, Sassani M, Ambrus A, Tretter L (2018) Membrane potential and delta pH dependency of reverse electron transport-associated hydrogen peroxide production in brain and heart mitochondria. J Bioenerg Biomembr 50:355-365 | 2018 | PMID: 30116920 Open Access |
| Krebs 1935 Biochem J | Krebs HA (1935) CXCVII. Metabolism of amino-acids. III. Deamination of amino-acids. Biochem J 29:1620-44. | 1935 | |
| Krebs 1940 Biochem J | Krebs HA (1940) The citric acid cycle. A reply to the criticisms of F. L. Breusch and of J. Thomas. Biochem J 34:460-3. | 1940 | PMID: 16747181 Open Access |
| Krogh 1929 Science | Krogh A (1929) The progress of physiology. Science 70:200–4. | 1929 | PMID: 17732865 |
| Krumschnabel 2014 Methods Enzymol | Krumschnabel G, Eigentler A, Fasching M, Gnaiger E (2014) Use of safranin for the assessment of mitochondrial membrane potential by high-resolution respirometry and fluorometry. Methods Enzymol 542:163-81. https://doi.org/10.1016/B978-0-12-416618-9.00009-1 | 2014 | PMID: 24862266 » |
| Kunz 2000 J Biol Chem | Kunz WS, Kudin A, Vielhaber S, Elger CE, Attardi G, Villani G (2000) Flux control of cytochrome c oxidase in human skeletal muscle. J Biol Chem 275:27741-5. | 2000 | PMID: 10869362 Open Access |
| Kuznetsov 1996 J Biol Chem | Kuznetsov AV, Clark JF, Winkler K, Kunz WS (1996) Increase of flux control of cytochrome c oxidase in copper-deficient mottled brindled mice. J Biol Chem 271:283-8. | 1996 | PMID: 8550574 |
| Kuznetsov 2004 Am J Physiol Heart Circ Physiol | Kuznetsov AV, Schneeberger S, Seiler R, Brandacher G, Mark W, Steurer W, Saks V, Usson Y, Margreiter R, Gnaiger E (2004) Mitochondrial defects and heterogeneous cytochrome c release after cardiac cold ischemia and reperfusion. Am J Physiol Heart Circ Physiol 286:H1633–41. | 2004 | PMID: 14693685 Open Access |
| Kuznetsov 2002 Anal Biochem | Kuznetsov AV, Strobl D, Ruttmann E, Königsrainer A, Margreiter R, Gnaiger E (2002) Evaluation of mitochondrial respiratory function in small biopsies of liver. Anal Biochem 305:186-94. | 2002 | PMID: 12054447 |
| Kuznetsov 1997 Biochim Biophys Acta | Kuznetsov AV, Winkler K, Kirches E, Lins H, Feistner H, Kunz WS (1997) Application of inhibitor titrations for the detection of oxidative phosphorylation defects in saponin-skinned muscle fibers of patients with mitochondrial diseases. Biochim Biophys Acta 1360:142-50. | 1997 | PMID: 9128179 Open Access |
| Koenig 1969 Biochem J | König T, Nicholls DG, Garland PB (1969) The inhibition of pyruvate and Ls(+)-isocitrate oxidation by succinate oxidation in rat liver mitochondria. Biochem J 114:589-96. | 1969 | Open Access |
| LaNoue 1972 J Biol Chem | LaNoue KF, Bryla J, Williamson JR (1972) Feedback interactions in the control of citric acid cycle activity in rat heart mitochondria. J Biol Chem 247:667-79. | 1972 | JBC Open Access |
| LaNoue 1979 Annu Rev Biochem | LaNoue KF, Schoolwerth AC (1979) Metabolite transport in mitochondria. Annu Rev Biochem 48:871-922. | 1979 | |
| Lane 2005 Oxford Univ Press | Lane N (2005) Power, sex, suicide: Mitochondria and the meaning of life. Oxford University Press. 354 pp. | 2005 | http://www.nick-lane.net |
| Laner 2014 Mitochondr Physiol Network MiP2014 | Laner V, Gnaiger E, eds (2014) Mitochondrial physiology – methods, concepts and biomedical perspectives. MiP2014. ISBN 978-3-9502399-9-7 | 2014 | Mitochondr Physiol Network 19.13:88 pp. |
| Lardy 1952 J Biol Chem | Lardy HA, Wellman H (1952) Oxidative phosphorylations; role of inorganic phosphate and acceptor systems in control of metabolic rates. J Biol Chem 195:215-24. | 1952 | PMID: 14938372 Open Access |
| Lehninger 1970 Worth | Lehninger AL (1970) Biochemistry. The molecular basis of cell structure and function. Worth:833 pp. | 1970 | |
| Lehar 2007 Molecular Systems Biol | Lehár J, Zimmermann GR, Krueger AS, Molnar RA, Ledell JT, Heilbut AM, Short GF, Giusti LC, Nolan GP, Magid OA, Lee MS, Borisy AA, Stockwell BR, Keith CT (2007) Chemical combination effects predict connectivity in biological systems. Molecular Systems Biol 3:80. | 2007 | |
| Lemasters 1984 J Biol Chem | Lemasters JJ (1984) The ATP-to-Oxygen Stoichiometries of Oxidative Phosphorylation by Rat Liver Mitochondria. J Biol Chem 259:13123-30. | 1984 | PMID: 6548475 |
| Lemieux 2017 Sci Rep | Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. doi:10.1038/s41598-017-02789-8 | 2017 | PMID: 28588260 Sci Rep Open Access |
| Lemieux 2011 Int J Biochem Cell Biol | Lemieux H, Semsroth S, Antretter H, Höfer D, Gnaiger E (2011) Mitochondrial respiratory control and early defects of oxidative phosphorylation in the failing human heart. Int J Biochem Cell Biol 43:1729–38. https://doi.org/10.1016/j.biocel.2011.08.008 | 2011 | PMID: 21871578 » |
| Lemieux 2019 bioRxiv | Lemieux H, Subarsky P, Doblander C, Wurm M, Troppmair J, Gnaiger E (2019) Mitochondrial respiratory function as an early biomarker of apoptosis induced by growth factor removal. bioRxiv doi: https://doi.org/10.1101/151480 . | 2019 | bioRxiv Preprint Version 1 Open Access Version 2 (2019-06-11) in preparation |
| Lenaz 2009 Int J Biochem Cell Biol | Lenaz G, Genova ML (2009) Structural and functional organization of the mitochondrial respiratory chain: A dynamic super-assembly. Int J Biochem Cell Biol 41:1750-72. | 2009 | PMID:19711505 Open Access |
| Llesuy 1994 Free Radic Biol Med | Llesuy S, Evelson P, González-Flecha B, Peralta J, Carreras MC, Poderoso JJ, Boveris A (1994) Oxidative stress in muscle and liver of rats with septic syndrome. Free Radic Biol Med 16:445-51. | 1994 | |
| Loomis 1948 J Biol Chem | Loomis WF, Lipmann F (1948) Reversible inhibition of the coupling between phosphorylation and oxidation. J Biol Chem 173:807-8. | 1948 | PMID: 18910739 Open Access |
| Maechler 2006 Int J Biochem Cell Biol | Maechler P, Carobbio S, Rubi B (2006) In beta-cells, mitochondria integrate and generate metabolic signals controlling insulin secretion. Int J Biochem Cell Biol 38:696-709. | 2006 | |
| Maxwell 1867 Phil Trans Royal Soc London | Maxwell JC ( 1867) On the dynamical theory of gases. Phil Trans Royal Soc London 157:49-88. | 1867 | File:Maxwell 1867 Phil Trans Royal Society London.pdf |
| Meredith 1983 Science | Meredith MA, Stein BE (1983) Interactions among converging sensory inputs in the superior colliculus. Science 221:389–91. | 1983 | |
| Messer 2004 Am J Physiol Cell Physiol | Messer JI, Jackman MR, Willis WT (2004) Pyruvate and citric acid cycle carbon requirements in isolated skeletal muscle mitochondria. Am J Physiol Cell Physiol 286:C565-72. | 2004 | PMID: 14602577 Open Access |
| Michel 2008 Proc Natl Acad Sci U S A | Michel JB, Yeh PJ, Chait R, Moellering RC Jr, Kishony R (2008) Drug interactions modulate the potential for evolution of resistance. Proc Natl Acad Sci U S A 105:14918-23. | 2008 | PMID: 18815368 Open Access |
| Mitchell 1961 Nature | Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 191:144-8. | 1961 | PMID: 13771349 |
| Mitchell 2011 Biochim Biophys Acta | Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. https://doi.org/10.1016/j.bbabio.2011.09.018 | 1966 | Biochim Biophys Acta Bioenergetics 1807 (2011):1507-38. Science Direct Open Access |
| Mitchell 1967 Nature | Mitchell P (1967) Proton current flow in mitochondrial systems. Nature 214:1327–8. | 1967 | https://www.nature.com/articles/2141327a0 |
| Mitchell 1979 Science | Mitchell P (1979) Keilin’s respiratory chain concept and its chemiosmotic consequences. Science 206:1148-59. | 1979 | |
| Mitchell 1967 Biochem J | Mitchell P, Moyle J (1967) Respiration-driven proton translocation in rat liver mitochondria. Biochem J 105:1147-62. | 1967 | PMID: 16742541 Open Access |
| Mogensen 2005 Acta Physiol Scand | Mogensen M, Sahlin K (2005) Mitochondrial efficiency in rat skeletal muscle: influence of respiration rate, substrate and muscle type. Acta Physiol Scand 185:229-36. | 2005 | |
| Mohr 2015 Metrologia | Mohr Peter J, Phillips William D (2015) Dimensionless units in the SI. Metrologia 52:40-7. https://doi.org/10.1088/0026-1394/52/1/40 | 2015 | Metrologia Open Access |
| Mootha 1997 Am J Physiol | Mootha VK, Arai AE, Balaban RS (1997) Maximum oxidative phosphorylation capacity of the mammalian heart. Am J Physiol 272:H769-775. | 1997 | PMID: 9124437 Open Access |
| Muller 2008 Biochem J | Muller FL, Liu Y, Abdul-Ghani MA, Lustgarten MS, Bhattacharya A, Jang YC, Van Remmen H (2008) High rates of superoxide production in skeletal-muscle mitochondria respiring on both Complex I- and Complex II-linked substrates. Biochem J 409:491–99. | 2008 | PMID: 17916065 Open Access |
| Natarajan 2006 Nature Cell Biol | Natarajan M, Lin KM, Hsueh C, Sternweis PC, Ranganathan R (2006) A global analysis of cross-talk in a mammalian cellular signalling network. Nature Cell Biol 8:571-80. | 2006 | |
| Nernst 1921 Nobel Lecture | Nernst W (1921) Studies in chemical thermodynamics. Nobel Lecture December 12, 1921:353-362. | 1921 | Nobel Lecture Open Access |
| Nicholls 2013 Academic Press | Nicholls DG, Ferguson SJ (2013) Bioenergetics4. Academic Press 419 pp. https://doi.org/10.1016/C2010-0-64902-9 | 2013 | |
| Nicholson 2012 Nature | Nicholson JK, Holmes E, Kinross JM, Darzi AW, Takats Z, Lindon JC (2012) Metabolic phenotyping in clinical and surgical environments. Nature 491:384-92. | 2012 | PMID: 23151581 |
| Odra Noel 2014 MiPNet | Odra Noel, Gnaiger E (2014) MiPArt - Mitchell's dream - Mitchell's equation. MiPNet 19.13:6-8. | 2014 | Laner and Gnaiger (2014) Mitochondr Physiol Network MiP2014 - Open Access |
| Onsager 1931 Phys Rev | Onsager L (1931) Reciprocal relations in irreversible processes. I. Phys Rev 37:405-26. https://doi.org/10.1103/PhysRev.37.405 | 1931 | Open Access |
| Ouhabi 1994 Modern Trends in BioThermoKinetics 3 | Ouhabi R, Boue-Grabot M, Mazat J-P (1994) ATP synthesis in permeabilized cells: Assessment of the ATP/O ratios in situ. Modern Trends in BioThermoKinetics 3 Innsbruck Univ Press. In What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds):141-4. | 1994 | |
| Owen 2002 J Biol Chem | Owen OE, Kalhan SC, Hanson RW (2002) The key role of anaplerosis and cataplerosis for citric acid cycle function. J Biol Chem 277:30409-12. | 2002 | PMID: 12087111 Open Access |
| O’Donnell 2004 Am J Physiol Heart Circ Physiol | O’Donnell JM, Kudej RK, LaNoue KF, Vatner SF, Lewandowski ED (2004) Limited transfer of cytosolic NADH into mitochondria at high cardiac workload. Am J Physiol Heart Circ Physiol 286:H2237-42. | 2004 | |
| Palade 1952 Anat Rec | Palade GE (1952) The fine structure of mitochondria. Anat Rec 114:427-51. | 1952 | PMID: 12996882 |
| Pardee 1948 J Biol Chem | Pardee AB, Potter VR (1948) Inhibition of succinic dehydrogenase by oxalacetate. J Biol Chem 176:1085-94. | 1948 | PMID: 18098561 |
| Patel 2008 J Med Chem | Patel Y, Gillet VJ, Howe T, Pastor J, Oyarzabal J, Willett P (2008) Assessment of additive/nonadditive effects in structure−activity relationships: Implications for iterative drug design. J Med Chem 51:7552–62. | 2008 | |
| Patzek 2014 Advances in Historical Studies | Patzek Tad W (2014) Fick’s diffusion experiments revisited — Part I. Advances in Historical Studies 3:194-206. | 2014 | Open Access |
| Pesta 2012 Methods Mol Biol | Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods Mol Biol 810:25-58. https://doi.org/10.1007/978-1-61779-382-0_3 | 2012 | PMID: 22057559 |
| Pesta 2011 Am J Physiol Regul Integr Comp Physiol | Pesta D, Hoppel F, Macek C, Messner H, Faulhaber M, Kobel C, Parson W, Burtscher M, Schocke M, Gnaiger E (2011) Similar qualitative and quantitative changes of mitochondrial respiration following strength and endurance training in normoxia and hypoxia in sedentary humans. Am J Physiol Regul Integr Comp Physiol 301:R1078–87. | 2011 | PMID: 21775647 Open Access |
| Pirsig 1974 William Morrow & Company | Pirsig RM (1974) Zen and the art of motorcycle maintenance. An inquiry into values. William Morrow & Company 418 pp. | 1974 | |
| Poburko 2011 J Biol Chem | Poburko Damon, Santo-Domingo Jaime, Demaurex Nicolas (2011) Dynamic regulation of the mitochondrial proton gradient during cytosolic calcium elevations. J Biol Chem 286:11672-84. | 2011 | Open Access |
| Ponsot 2005 J Cell Physiol | Ponsot E, Zoll J, N'guessan B, Ribera F, Lampert E, Richard R, Veksler V, Ventura-Clapier R, Mettauer B (2005) Mitochondrial tissue specificity of substrates utilization in rat cardiac and skeletal muscles. J Cell Physiol 203:479-86. | 2005 | PMID: 15521069 |
| Prebble 2003 Oxford Univ Press | Prebble J, Weber B (2003) Wandering in the gardens of the mind. Peter Mitchell and the making of Glynn. Oxford Univ Press Oxford. | 2003 | |
| Prigogine 1967 Interscience | Prigogine I (1967) Introduction to thermodynamics of irreversible processes. Interscience, New York, 3rd ed:147pp. | 1967 | |
| Puchowicz 2004 Mitochondrion | Puchowicz MA, Varnes ME, Cohen BH, Friedman NR, Kerr DS, Hoppel CL (2004) Oxidative phosphorylation analysis: assessing the integrated functional activity of human skeletal muscle mitochondria-case studies. Mitochondrion 4:377-85. | 2004 | PMID: 16120399 |
| Rabinovitz 1951 Science | Rabinovitz M, Stulberg MP, Boyer PD (1951) The control of pyruvate oxidation in a cell-free rat heart preparation by phosphate acceptors. Science 114:641-2. | 1951 | PMID: 14913129 |
| Raboel 2009 J Clin Endocrinol Metabol | Rabøl R, Højberg PM, Almdal T, Boushel R, Haugaard SB, Madsbad S, Dela F (2009) Effect of hyperglycemia on mitochondrial respiration in type 2 diabetes. J Clin Endocrinol Metabol 94:1372-78. | 2009 | PMID: 19141588 ; Open Access |
| Rasmussen 1997 Mol Cell Biochem | Rasmussen HN, Rasmussen UF (1997) Small scale preparation of skeletal muscle mitochondria, criteria of integrity, and assays with reference to tissue function. Mol Cell Biochem 174:55-60. | 1997 | PMID: 9309665 |
| Rasmussen 2000 Mol Cell Biochem | Rasmussen UF, Rasmussen HN (2000) Human quadriceps muscle mitochondria: a functional characterization. Mol Cell Biochem 208:37-44. | 2000 | PMID: 10939626 |
| Rasmussen 1996 Biochem J | Rasmussen UF, Rasmussen HN, Andersen AJ, Fogd Jørgensen P, Quistorff B. (1996) Characterization of mitochondria from pig muscle: higher activity of exo-NADH oxidase in animals suffering from malignant hyperthermia. Biochem J 315:659-63. | 1996 | PMID: 8615844 |
| Rasmussen 2001 Am J Physiol Endocrinol Metab | Rasmussen UF, Rasmussen HN, Krustrup P, Quistorff B, Saltin B, Bangsbo J (2001) Aerobic metabolism of human quadriceps muscle: in vivo data parallel measurements on isolated mitochondria. Am J Physiol Endocrinol Metab 280:E301-7. | 2001 | PMID: 11158934 |
| Rasmussen 2004 Comp Biochem Physiol | Rasmussen UF, Vielwerth SE, Rasmussen HN (2004) Skeletal muscle bioenergetics: a comparative study of mitochondria isolated from pigeon pectoralis, rat soleus, rat biceps brachii, pig biceps femoris and human quadriceps. Comp Biochem Physiol A Molec Integr Physiol 137:435-46. | 2004 | PMID:15123217 |
| Rauchova 2003 Acta Biochim Polonica | Rauchova H, Drahota Z, Rauch P, Fato R, Lenaz G (2003) Coenzyme Q releases the inhibitory effect of free fatty acids on mitochondrial glycerophosphate dehydrogenase. Acta Biochim Polonica 50:405-13. | 2003 | |
| Renner 2003 Biochim Biophys Acta | Renner K, Amberger A, Konwalinka G, Gnaiger E (2003) Changes of mitochondrial respiration, mitochondrial content and cell size after induction of apoptosis in leukemia cells. Biochim Biophys Acta 1642:115-23. | 2003 | PMID: 12972300 Open Access |
| Rich 2003 Nature | Rich P (2003) Chemiosmotic coupling: The cost of living. Nature 421:583. | 2003 | PMID: 12571574 |
| Rossignol 2003 Biochem J | Rossignol R, Faustin B, Rocher C, Malgat M, Mazat JP, Letellier T (2003) Mitochondrial threshold effects. Biochem J 370:751-62. | 2003 | PMID:12467494 |
| Rottenberg 1984 J Membr Biol | Rottenberg H (1984) Membrane potential and surface potential in mitochondria: uptake and binding of lipophilic cations. J Membr Biol 81:127-38. | 1984 | PMID:6492133 |
| Rumsey 1990 J Biol Chem | Rumsey WL, Schlosser C, Nuutinen EM, Robiolio M, Wilson DF (1990) Cellular energetics and the oxygen dependence of respiration in cardiac myocytes isolated from adult rat. J Biol Chem 265:15392-9. | 1990 | |
| Saks 1998 Mol Cell Biochem | Saks VA, Veksler VI, Kuznetsov AV, Kay L, Sikk P, Tiivel T, Tranqui L, Olivares J, Winkler K, Wiedemann F, Kunz WS (1998) Permeabilised cell and skinned fiber techniques in studies of mitochondrial function in vivo. Mol Cell Biochem 184:81-100. | 1998 | PMID: 9746314 |
| Sauer 1973 FEBS Lett | Sauer LA (1973) Mitochondrial NAD-dependent malic enzyme: a new regulatory enzyme. FEBS Lett 33:251-5. | 1973 | PMID: 4147096 Open Access |
| Scaduto 1999 Biophys J | Scaduto RC Jr, Grotyohann LW (1999) Measurement of mitochondrial membrane potential using fluorescent rhodamine derivatives. Biophys J 76:469-77. | 1999 | PMID:9876159 Open Access |
| Schatz 2007 Annu Rev Biochem | Schatz G (2007) The magic garden. Annu Rev Biochem 76: 673-678. | 2007 | PMID: 17313357 Open Access |
| Schrödinger 1944 Cambridge Univ Press | Schrödinger E (1944) What is life? The physical aspect of the living cell. Cambridge Univ Press (1st edition). | 1944 | |
| Schwerzmann 1986 J Cell Biol | Schwerzmann K, Cruz-Orive LM, Eggman R, Sänger A, Weibel ER (1986) Molecular architecture of the inner membrane of mitochondria from rat liver: a combined biochemical and stereological study. J Cell Biol 102:97-103. | 1986 | PMID: 2867101 Open Access |
| Schwerzmann 1989 Proc Natl Acad Sci U S A | Schwerzmann K, Hoppeler H, Kayar SR, Weibel ER (1989) Oxidative capacity of muscle and mitochondria: correlation of physiological, biochemical, and morphometric characteristics. Proc Natl Acad Sci U S A 86:1583-7. | 1989 | Open Access |
| Schaegger 2001 J Biol Chem | Schägger H, Pfeiffer K (2001) The ratio of oxidative phosphorylation complexes I-V in bovine heart mitochondria and the composition of respiratory chain supercomplexes. J Biol Chem 276:37861-7. | 2001 | PMID: 11483615 Open Access |
| Schoepf 2020 Nat Commun | Schöpf Bernd, Weissensteiner Hansi, Schäfer Georg, Fazzini Federica, Charoentong Pornpimol, Naschberger Andreas, Rupp Bernhard, Fendt Liane, Bukur Valesca, Giese Irina, Sorn Patrick, Sant’Anna-Silva Ana Carolina, Iglesias-Gonzalez Javier, Sahin Ugur, Kronenberg Florian, Gnaiger Erich, Klocker Helmut (2020) OXPHOS remodeling in high-grade prostate cancer involves mtDNA mutations and increased succinate oxidation. https://doi.org/10.1038/s41467-020-15237-5 | 2020 | Nat Commun 11:1487. PMID: 32198407 Open Access » |
| Segre 2005 Nat Genet | Segrè D, DeLuna A, Church GM, Kishony R (2005) Modular epistasis in yeast metabolism. Nat Genet 37:77–83. | 2005 | |
| Short 2005 Proc Natl Acad Sci U S A | Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS (2005) Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A 102:5618-23. | 2005 | PMID: 15800038 Open Access |
| Simon 2008 Biochim Biophys Acta | Simon J, van Spanning RJM, Richardson DJ (2008) The organisation of proton motive and non-proton motive redox loops in prokaryotic respiratory systems. Biochim Biophys Acta 1777:1480–90. | 2008 | |
| Singh 1997 Fourth Estate | Singh Simon (1997) Fermat's last theorem. Fourth Estate, London 340 pp. | 1997 | http://www.simonsingh.com |
| Slater 1952 Nature | Slater EC, Cleland KW (1952) Stabilization of oxidative phosphorylation in heart-muscle sarcosomes. Nature 170:118-9. | 1952 | PMID: 14957042 |
| Slater 1973 Biochim Biophys Acta | Slater EC, Rosing J, Mol A (1973) The phosphorylation potential generated by respiring mitochondria. Biochim Biophys Acta 292:534-53. | 1973 | |
| Stadlmann 2006 Cell Biochem Biophys | Stadlmann S, Renner K, Pollheimer J, Moser PL, Zeimet AG, Offner FA, Gnaiger E (2006) Preserved coupling of oxidative phosphorylation but decreased mitochondrial respiratory capacity in IL-1ß treated human peritoneal mesothelial cells. Cell Biochem Biophys 44:179-86. | 2006 | PMID: 16456220 |
| Stadlmann 2002 Transplantation | Stadlmann S, Rieger G, Amberger A, Kuznetsov AV, Margreiter R, Gnaiger E (2002) H2O2-mediated oxidative stress versus cold ischemia-reperfusion: mitochondrial respiratory defects in cultured human endothelial cells. Transplantation 74:1800-3. | 2002 | PMID: 12499903 |
| Steinlechner-Maran 1996 Am J Physiol Cell Physiol | Steinlechner-Maran R, Eberl T, Kunc M, Margreiter R, Gnaiger E (1996) Oxygen dependence of respiration in coupled and uncoupled endothelial cells. Am J Physiol Cell Physiol 271:C2053-61. https://doi.org/10.1152/ajpcell.1996.271.6.C2053 | 1996 | PMID: 8997208 |
| Steinlechner-Maran 1997 Transplantation | Steinlechner-Maran R, Eberl T, Kunc M, Schröcksnadel H, Margreiter R, Gnaiger E (1997) Respiratory defect as an early event in preservation/reoxygenation injury in endothelial cells. Transplantation 63:136-42. | 1997 | PMID: 9000675 Open Access |
| Stock 1999 Science | Stock D, Leslie AGW, Walker JE (1999) Molecular architecture of the rotary motor in ATP synthase. Science 286:1700-05. | 1999 | |
| Sugano 1974 Biochim Biophys Acta | Sugano T, Oshino N, Chance B (1974) Mitochondrial functions under hypoxic conditions. The steady states of cytochrome c reduction and energy metabolism. Biochim Biophys Acta 347:340-58. | 1974 | |
| Sun 2005 Cell | Sun F, Huo X, Zhai Y, Wang A, Xu J, Su D, Bartlam M, Rao Z (2005) Crystal structure of mitochondrial respiratory membrane protein Complex II. Cell 121:1043–57. https://doi.org/10.1016/j.cell.2005.05.025 | 2005 | PMID: 15989954 Open Access |
| Swenson 2018 Anesthesiology | Swenson ER (2018) Does aerobic respiration produce carbon dioxide or hydrogen ion and bicarbonate? https://doi.org/10.1097/ALN.0000000000002125 | 2018 | Anesthesiology 128:873–9. Open Access |
| Territo 2000 Am J Physiol Cell Physiol | Territo PR, Mootha VK, French SA, Balaban RS (2000) Ca2+ activation of heart mitochondrial oxidative phosphorylation: role of the F0/F1-ATPase. Am J Physiol Cell Physiol 278:C423-35. | 2000 | |
| Tonkonogi 1999 Eur J Physiol | Tonkonogi M, Walsh B, Tiivel T, Saks V, Sahlin K (1999) Mitochondrial funciton in human skeletal muscle is not impaired by high intensity exercise. Eur J Physiol 437:562-8. | 1999 | |
| Torella 2010 PLoS Comput Biol | Torella JP, Chait R, Kishony R (2010) Optimal drug synergy in antimicrobial treatments. PLoS Comput Biol 6:e1000796. doi:10.1371/journal.pcbi.1000796. | 2010 | |
| Torres 1988 Int J Biochem | Torres NV, Mateo F, Sicilia J, Meléndez-Hevia E (1988) Distribution of the flux control in convergent metabolic pathways: theory and application to experimental and simulated systems. Int J Biochem 20:161-165. | 1988 | |
| Tretiakov 2009 J Phys Chem B | Tretiakov KV, Bishop KJM, Grzybowski BA (2009) Additivity of the excess energy dissipation rate in a dynamically self-assembled system. J Phys Chem B 113:7574–8. | 2009 | |
| Van't Hoff 1901 Nobel Lecture | Van't Hoff JH (1901) Osmotic pressure and chemical equilibrium. Nobel Lecture December 13, 1901:5-10. | 1901 | Nobel Lecture Open Access |
| Villani 1997 Proc Natl Acad Sci U S A | Villani G, Attardi G (1997) In vivo control of respiration by cytochrome c oxidase in wild-type and mitochondrial DNA mutation-carrying human cells. Proc Natl Acad Sci U S A 94:1166-71. | 1997 | PMID: 9037024 Open Access |
| Villani 1998 J Biol Chem | Villani G, Greco M, Papa S, Attardi G (1998) Low reserve of cytochrome c oxidase capacity in vivo in the respiratory chain of a variety of human cell types. J Biol Chem 273:31829-36. | 1998 | PMID: 9822650 Open Access |
| Votion 2012 PLoS One | Votion DM, Gnaiger E, Lemieux H, Mouithys-Mickalad A, Serteyn D (2012) Physical fitness and mitochondrial respiratory capacity in horse skeletal muscle. PLoS One 7:e34890. https://doi.org/10.1371/journal.pone.0034890 | 2012 | PMID: 22529950 Open Access |
| Wallace 2010 Annu Rev Pathol | Wallace DC, Fan W, Procaccio V (2010) Mitochondrial energetics and therapeutics. Annu Rev Pathol 5:297-348. | 2010 | PMID: 20078222 |
| Wang 2010 Phys Rev D | Wang T (2010) Coulomb force as an entropic force. Phys Rev D 81:104045. | 2010 | Phys Rev D Open Access arXiv |
| White 1997 Fourth Estate | White M (1997) Isaak Newton. The last sorcerer. Fourth Estate, London 402 pp. | 1997 | |
| Wiedemann 1998 J Neurol Sci | Wiedemann FR, Winkler K, Kuznetsov AV, Bartels C, Vielhaber S, Feistner H, Kunz WS (1998) Impairment of mitochondrial function in skeletal muscle of patients with amyotrophic lateral sclerosis. J Neurol Sci 156:65-72. | 1998 | PMID: 9559989 |
| Wilson 1988 J Biol Chem | Wilson DF, Rumsey WL, Green TJ, Vanderkooi J (1988) The oxygen dependence of mitochondrial oxidative phosphorylation measured by a new optical method for measuring oxygen concentration. J Biol Chem 263:2712-8. | 1988 | |
| Winkler-Stuck 2005 J Neural Transm | Winkler-Stuck K, Kirches E, Mawrin C, Dietzmann K, Lins H, Wallesch CW, Kunz WS, Wiedemann FR (2005) Re-evaluation of the dysfunction of mitochondrial respiratory chain in skeletal muscle of patients with Parkinson's disease. J Neural Transm 112:499-518. | 2005 | PMID: 15340872 |
| Yeh 2006 Nat Genet | Yeh P, Tschumi AI, Kishony R (2006) Functional classification of drugs by properties of their pairwise interactions. Nat Genet 38:489-94. | 2006 | |
| Yeh 2009 Nat Rev Microbiol | Yeh PJ, Hegreness MJ, Aiden AP, Kishony R (2009) Drug interactions and the evolution of antibiotic resistance. Nat Rev Microbiol 7:460–6. | 2009 | |
| Zoccarato 2007 Biochem J | Zoccarato F, Cavallini L, Bortolami S, Alexandre A (2007) Succinate modulation of H2O2 release at NADH:ubiquinone oxidoreductase (Complex I) in brain mitochondria. Biochem J 406:125–9. | 2007 | |
| Kemp 1994 BTK-226 | Kemp RB, Hoare S, Schmalfeldt M, Bridge ChMC, Evansa PM, Gnaiger E (1994) A thermochemical study of the producion of lactate by glutaminolysis and glycolysis in mouse macrophage hybridoma cells. In: What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds) Modern Trends in BioThermoKinetics 3. Innsbruck Univ Press:226-31. | 1994 | |
| Gnaiger 1994 BTK-62 | Gnaiger E (1994) Negative entropy for living systems: controversy between Nobel Laureates Schrödinger, Pauling and Perutz. In: What is Controlling Life? (Gnaiger E, Gellerich FN, Wyss M, eds) Modern Trends in BioThermoKinetics 3. Innsbruck Univ Press: 62-70. | 1994 |
BEC tutorial-Living Communications
Cited by
- Lane Nick (2022) Transformer: the deep chemistry of life and death. Profile Books:400 pp. ISBN-10: 0393651487 - »Bioblast link«
- Under 'Further reading': Erich Gnaiger, Mitochondrial Pathways and Respiratory Control. An Introduction to OXPHOS Analysis (Innsbruck, Bioenergetics Communications, 2020). Available here: http://doi:10.26124/bec:2020-0002. The 'bible' of fluorespirometry, privately published by Erich Gnaiger in the tradition of Peter Mitchell's 'little grey books'; this is the little blue book. Gives practical insights into how the Krebs cycle really works. Introduces the idea of the Q junction, where electrons funnel from many substrates, including glycerol phosphate outside the mitochondria, into Complex III.
- Gnaiger E, Cardoso LHD, Tindle-Solomon L, Cocco P, eds (2022) Bioblast 2022: BEC inaugural conference. https://doi.org/10.26124/bec:2022-0001
- Heimler SR, Phang HJ, Bergstrom J, Mahapatra G, Dozier S, Gnaiger E, Molina AJA (2021) Platelet bioenergetics are associated with resting metabolic rate and exercise capacity in older adult women. https://doi.org/10.26124/bec:2022-0002
- Zdrazilova L, Hansikova H, Gnaiger E (2022) Comparable respiratory activity in attached and suspended human fibroblasts. https://doi.org/10.1371/journal.pone.0264496
- Komlódi et al (2022) The protonmotive force - not merely membrane potential. MitoFit Preprints 2022 (in prep)
- Donnelly C, Schmitt S, Cecatto C, Cardoso LHD, Komlodi T, Place N, Kayser B, Gnaiger E (2022) The ABC of hypoxia – what is the norm. https://doi.org/10.26124/mitofit:2022-0025
- Baglivo E, Cardoso LHD, Cecatto C, Gnaiger E (2022) Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry. https://doi.org/10.26124/mitofit:2022-0018.v2
- Gnaiger E (2021) Beyond counting papers – a mission and vision for scientific publication. Bioenerg Commun 2021.5. https://doi:10.26124/BEC:2021-0005
- Vernerova A, Garcia-Souza LF, Soucek O, Kostal M, Rehacek V, Krcmova LK, Gnaiger E, Sobotka O (2021) Mitochondrial respiration of platelets: comparison of isolation methods. https://doi.org/10.3390/biomedicines9121859
- Gnaiger E (2021) Beyond counting papers – a mission and vision for scientific publication. Bioenerg Commun 2021.5. https://doi:10.26124/BEC:2021-0005
- Gnaiger E (2021) Bioenergetic cluster analysis – mitochondrial respiratory control in human fibroblasts. MitoFit Preprints 2021.8. https://doi.org/10.26124/mitofit:2021-0008
- Krako Jakovljevic N, Ebanks B, Katyal G, Chakrabarti L, Markovic I, Moisoi N (2021) Mitochondrial homeostasis in cellular models of Parkinson’s Disease. Bioenerg Commun 2021.2. https://doi.org/10.26124/bec:2021-0002
- Komlódi T, Cardoso LHD, Doerrier C, Moore AL, Rich PR, Gnaiger E (2021) Coupling and pathway control of coenzyme Q redox state and respiration in isolated mitochondria. Bioenerg Commun 2021.3. https://doi.org/10.26124/bec:2021-0003
- Cardoso et al (2021) Magnesium Green for fluorometric measurement of ATP production does not interfere with mitochondrial respiration. Bioenerg Commun 2021.1. doi:10.26124/bec:2021-0001
- Went N, Di Marcello M, Gnaiger E (2021) Oxygen dependence of photosynthesis and light-enhanced dark respiration studied by High-Resolution PhotoRespirometry. MitoFit Prep 2021.5. - »Bioblast link«
- Silva et al (2021) Off-target effect of etomoxir on mitochondrial Complex I. MitoFit Preprints 2021. (in preparation)
- Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. doi:10.26124/bec:2020-0001.v1.
- Gnaiger E (2021) Bioenergetic cluster analysis – mitochondrial respiratory control in human fibroblasts. MitoFit Preprints 2021.8. https://doi.org/10.26124/mitofit:2021-0008
Labels: MiParea: Respiration, Instruments;methods, mt-Biogenesis;mt-density, Comparative MiP;environmental MiP, Exercise physiology;nutrition;life style, mt-Medicine, mt-Awareness
Organism: Human, Mouse
Tissue;cell: Heart, Skeletal muscle, Fibroblast
Preparation: Permeabilized cells, Permeabilized tissue, Homogenate, Isolated mitochondria, Intact cells
Regulation: Coupling efficiency;uncoupling, Flux control, mt-Membrane potential, Threshold;excess capacity, Uncoupler Coupling state: LEAK, ROUTINE, OXPHOS, ET Pathway: F, N, S, Gp, CIV, NS, ROX HRR: Oxygraph-2k, O2k-Fluorometer, O2k-Protocol, Theory
Additivity, Ambiguity crisis, BEC, BEC2020, MitoFitPublication, MitoEAGLEPublication, MitoPathways, O2k-chemicals and media, Mt-preparations, O2k-Demo, O2k-Core, 1R;2Omy;3U-, 1PGM;2D;3U-, 1PGM;2D;2c;3S;4U;5Rot;6Ama, 1OctM;2D;3G;4S;5U;6Rot;7Ama, Activity, Advancement, Amount of substance, Ampere, Assay, Avogadro constant, Barometric pressure, Biochemical coupling efficiency, Body mass, Boltzmann constant, Cell count and normalization in HRR, Cell respiration, Charge number, Citrate synthase activity, Closed chamber, Concentration, Count, Coupling-control ratio, Density, Dimension, Efficiency, Electric current, Electrochemical constant, Electron transfer pathway, Elementary charge, Elementary entity, Energy, Entity, ET capacity, Extensive quantity, E-L coupling efficiency, E-L net ET capacity, E-P excess capacity, E-P control efficiency, E-R reserve capacity, E-R control efficiency, Elementary charge, Faraday constant, Flow, Flux, Flux control efficiency, Flux control ratio, Force, Format, Gas constant, High-resolution respirometry, Iconic symbols, International System of Units, Isolated mitochondria, LEAK-respiration, Living cells, L/P coupling control ratio, L/R coupling control ratio, Malic enzyme, Mass, Mitochondrial marker, Mole, Motive unit, Normalization of rate, Number, Open chamber, OXPHOS capacity, OXPHOS-control ratio, Oxygen flow, Oxygen pressure, Oxygen solubility, Power, P-L net OXPHOS capacity, Pascal, Pathway control ratio, Permeabilized cells, Pressure, P-L control efficiency, Pressure, Protonmotive force, Quantities, Quantity, Reproducibility crisis, Residual oxygen consumption, Respiratory states, Respirometry, ROUTINE-control ratio, ROUTINE-coupling efficiency R-L, ROUTINE respiration, R-L net ROUTINE capacity, Sample, SI prefixes, Solubility, Solutions, Specific quantity, Symbols, System, Unit, Volume, Work, BEC 2020.1, X-mass Carol, MitoFit 2021 MgG, MitoFit 2021 CoQ, MitoFit 2021.5 PB, MitoFit 2021 BCA, MitoFit 2021 PLT, BEC2021.5, PLoSONE2022ace-sce, MitoFit2022Hypoxia, MitoFit 2022 NADH, MitoFit 2022 pmF, MitoFit2022QC
MitoPedia topics:
BEC